Why Veins Don't Get Heart Disease?
Feb 14, 2026
Why Arteries Get Atherosclerosis and Not Veins?
Many people online ask this question. If LDL particles trafficking cholesterol cause atherosclerosis, why don’t veins get atherosclerosis? If LDL-C is the cause, and LDL particles are circulating in plasma, even on the venous side, why don’t veins get atherosclerosis?
Smart question, right?
No. This is another case of these Medfluencers just not knowing enough. Unfortunately, sometimes doctors fall for this too.
Arteries And Veins Are Structurally Very Different
Arteries and veins are very different. Arteries are thicker, they have multiple layers, they have a muscle layer. They are not veins. Veins are thinner and composed of very different materials. They are not interchangeable. Arteries are also a higher-pressure system, which can drive lipoproteins into the intimal layer.
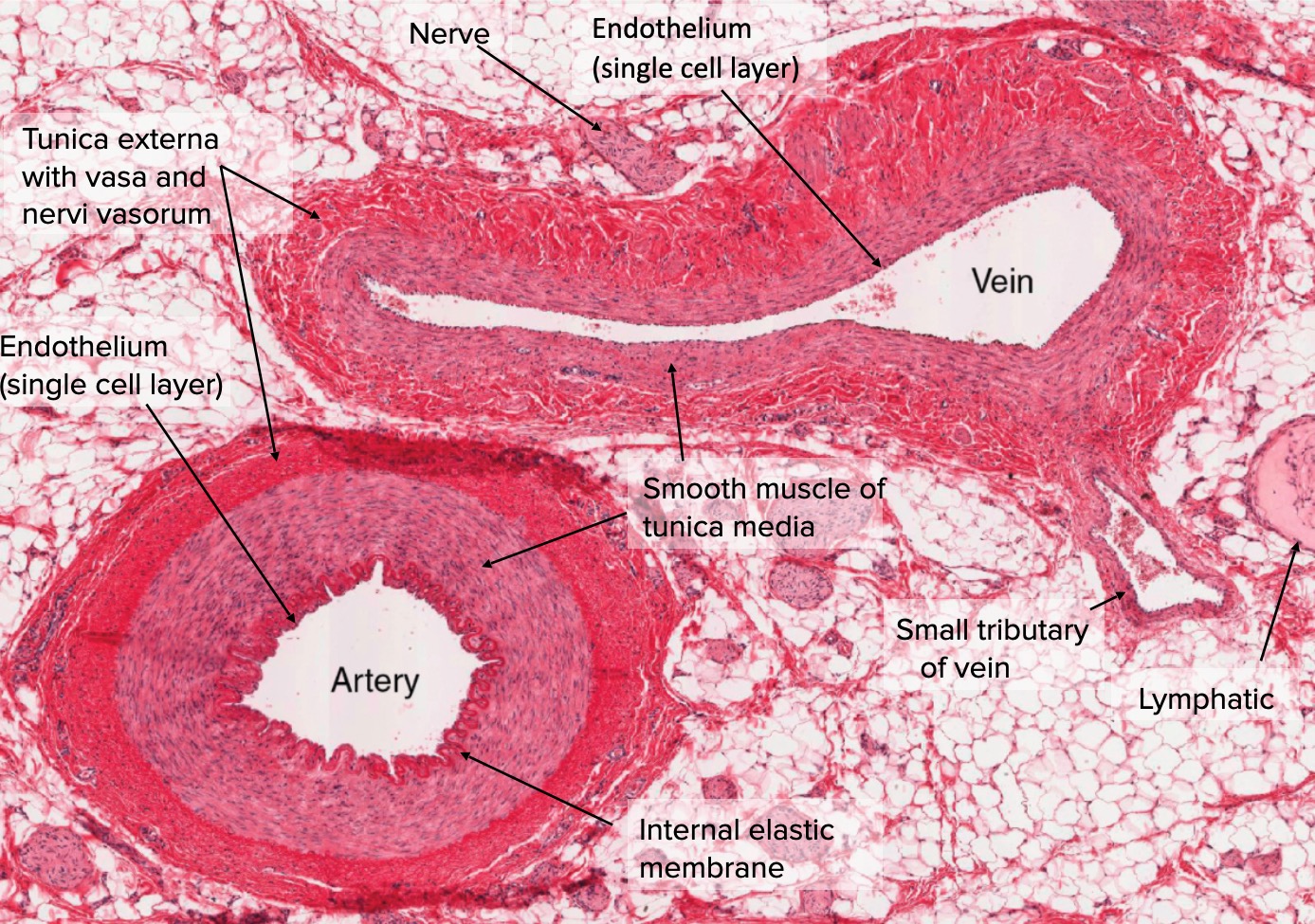
The above is a slide of an artery and a vein side by side under a microscope. Notice the vein is collapsed and the artery is held open. The artery is a higher-pressure system and has muscle, which helps keep it open. It’s more robust and has more layers than a vein. Medfluencers like to deny this fact and use cartoon models to illustrate how similar they are. They really aren’t.
Why Veins Don't Get Atherosclerosis: The Science Behind Vascular Disease Patterns
If you've ever wondered why heart attacks and strokes involve blocked arteries but not veins, you're asking one of the most fundamental questions in cardiovascular medicine. Despite being exposed to the same blood, the same cholesterol levels, and the same risk factors like smoking and diabetes, veins almost never develop atherosclerosis. The answer lies in a fascinating combination of physics, biology, and developmental programming.
The Pressure Difference
The most obvious difference between arteries and veins is pressure. Arteries operate under high pressure, your blood pressure reading of 120/80 mmHg reflects the force in your arteries. Veins, by contrast, operate in a low-pressure system, typically under 10 mmHg.
But it's not just about high versus low pressure. It's about the specific patterns of blood flow and the forces they create on vessel walls. Scientists call this force "shear stress", essentially the friction of blood flowing against the vessel lining.
Here's where it gets interesting: atherosclerosis doesn't develop in areas of high shear stress. Instead, plaques form precisely where blood flow becomes turbulent, slow, or oscillating, typically at branch points and curves in arteries. Studies have measured shear stress at these vulnerable spots at around ±4 dyne/cm², compared to greater than 12 dyne/cm² in protected arterial regions.
Veins experience uniformly low shear stress throughout. Paradoxically, this consistent low-pressure, low-shear environment may actually protect them from the inflammatory cascade that initiates atherosclerosis in arteries.
These differences have profound biological consequences. When endothelial cells, the single-cell layer lining all blood vessels, experience high laminar shear stress, they activate protective genetic programs. They decrease expression of vasoconstrictors, inflammatory mediators, and adhesion molecules while increasing production of vasodilators, fibrinolytics, and antioxidants. This creates an atheroprotective phenotype that actively resists plaque formation.
The protective effect of high shear stress explains a remarkable observation: even within arteries, disease distribution is highly focal. Despite systemic risk factors like hypercholesterolemia, smoking, and diabetes affecting the entire vasculature, plaques develop preferentially at vessel branch points, curvatures, and bifurcations, precisely the locations where blood flow becomes disturbed and shear stress drops to low or oscillatory levels (0±4 dynes/cm²). Straight arterial segments with high laminar flow remain plaque-free, even in patients with severe atherosclerosis elsewhere.
This focal distribution demonstrates that local hemodynamic forces, not just systemic factors, determine atherosclerosis susceptibility. The disease develops where protection fails, not uniformly throughout the arterial tree.
Arteries And Veins Are Not The Same Infographic Summary:
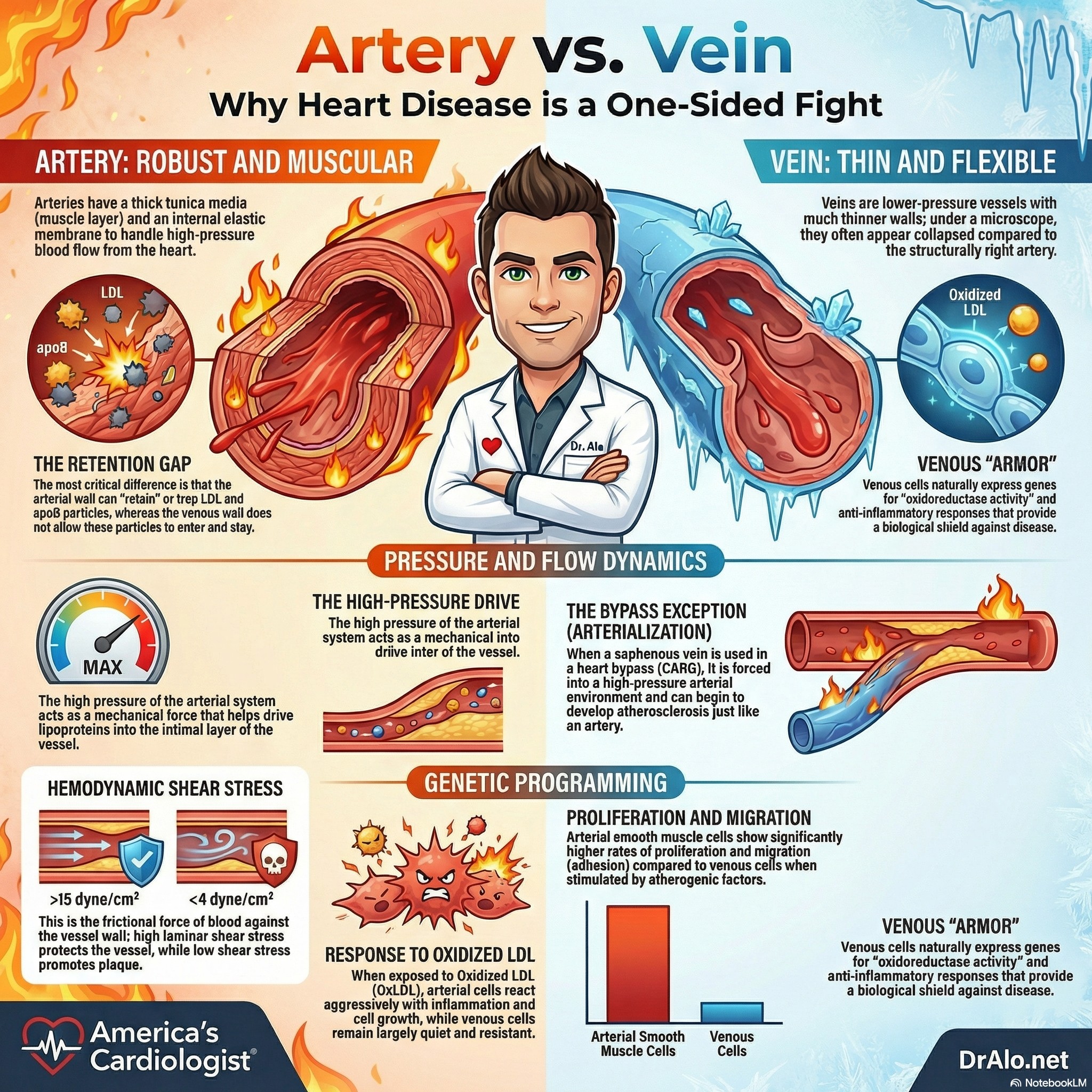
Arteries and Veins Are Built Differently From the Start
Even before birth, your body knows which blood vessels will become arteries and which will become veins. During embryonic development, specific genes activate in arterial cells (like Notch1/4 and EphrinB2) while different genes activate in venous cells (like COUP-TFII and EphB4). This isn't just a labeling system, it fundamentally programs how these cells will behave for the rest of your life.
When researchers exposed arterial and venous endothelial cells (the cells lining blood vessels) to oxidized LDL, one of the key triggers of atherosclerosis, the responses were dramatically different. Arterial cells showed massive activation of genes related to inflammation, cell adhesion, proliferation, and cell death. Venous cells? Barely a response.
This suggests that even if you could somehow expose veins to arterial-level pressure and cholesterol, they might still resist atherosclerosis because they're molecularly programmed differently.
Arterial vs. Venous Cell Identity At the Molecular and Cellular Level
Beyond hemodynamics, arteries and veins are fundamentally different at the cellular and molecular level. During embryonic development, arterial and venous endothelial cells are genetically programmed along distinct pathways. Arterial cells express specific markers including Notch1/4, Dll4, and EphrinB2, while venous cells express COUP-TFII and EphB4. These aren't just labels, they represent different functional programs.
When researchers expose coronary artery endothelial cells and saphenous vein endothelial cells to the same atherogenic stimuli, such as reactive oxygen species, oxidized LDL, the responses differ dramatically. Arterial cells show far greater activation of genes related to adhesion, proliferation, and apoptosis pathways. Similarly, smooth muscle cells harvested from atherosclerotic arteries demonstrate significantly greater proliferation, adhesion, and migration capacity compared to venous smooth muscle cells.
These intrinsic differences suggest that arterial cells are inherently more reactive to atherogenic stimuli, they're primed to respond more vigorously to injury and inflammation. This may reflect evolutionary adaptation to the high-stress arterial environment, but it also makes arteries more vulnerable when that environment becomes pathological.
The Smooth Muscle Story
The cells beneath the vessel lining, vascular smooth muscle cells, also behave differently depending on whether they come from arteries or veins. When scientists compared smooth muscle cells from atherosclerotic arteries to those from veins, the arterial cells showed significantly greater proliferation, adhesion, and migration.
These are exactly the cellular behaviors that drive the formation of atherosclerotic plaques. The arterial smooth muscle cells seem primed for the hyperplastic (overgrowth) responses that characterize atherosclerosis, while venous cells remain relatively quiescent.
What Happens When Veins Face Arterial Conditions?
There's a natural experiment that tests these theories: vein grafts used in coronary bypass surgery (CABG). When surgeons use a saphenous vein from your leg to bypass a blocked coronary artery, that vein suddenly experiences arterial pressures and flow patterns.
And what happens? The vein often develops atherosclerosis-like disease over time. This suggests that while veins have some intrinsic protection, hemodynamic forces play a crucial role. The combination of low pressure and intrinsic cellular resistance likely explains why veins in their natural environment remain disease-free.
When surgeons harvest a saphenous vein from the leg and use it to bypass a blocked coronary artery, they transplant a vein into the arterial circulation. What happens next is striking.
Within the first year after surgery, up to 20% of saphenous vein grafts become totally occluded. By 10 years, approximately 40% are completely blocked, and an additional 50% show advanced atherosclerotic changes. The same veins that remained healthy for decades in the low-pressure venous system rapidly develop disease when exposed to arterial conditions.
The mechanisms driving this transformation are initially distinct from native arterial atherosclerosis. Venous tissue suddenly exposed to high arterial pressures undergoes structural remodeling characterized by smooth muscle cell proliferation, neointimal hyperplasia (thickening of the inner vessel layer), and extracellular matrix expansion. However, after the first 1-2 years, the pathological changes in vein grafts become indistinguishable from those seen in native arterial atherosclerosis, complete with cholesterol-laden plaques, inflammatory cell infiltration, and calcification.
This transformation reveals a crucial insight: venous endothelial cells are physiologically adapted to chronic low shear stress. When suddenly exposed to acute high arterial shear stress after grafting, they don't activate the protective programs that native arterial cells would. Instead, they activate pro-inflammatory profiles mediated by MAPK and NF-κB signaling pathways. The same cells that were protected in the venous environment become atherosclerosis-prone when placed in arterial conditions.
The Protective Paradox: Why High Flow Protects
The relationship between shear stress and atherosclerosis seems counterintuitive at first. One might expect that higher mechanical forces would cause more damage. Instead, the opposite is true: high laminar shear stress protects, while low or oscillatory shear stress promotes disease.
The explanation lies in how endothelial cells sense and respond to mechanical forces. These cells contain mechanosensors, protein complexes that detect fluid shear stress and convert mechanical signals into biochemical responses. Under high laminar flow, these sensors activate protective pathways that maintain endothelial integrity, suppress inflammation, and prevent LDL infiltration into the vessel wall.
Under low or oscillatory flow, the same sensors trigger opposite responses. Endothelial cells become activated and dysfunctional, expressing adhesion molecules that recruit inflammatory cells, increasing permeability to allow LDL penetration, and producing reactive oxygen species that oxidize LDL particles. This creates the perfect environment for atherosclerosis initiation.
Veins, operating under chronically low shear stress, might be expected to develop these pathological changes. However, venous endothelial cells are adapted to this environment—they're programmed to function normally under low flow conditions. The problem arises when vessels experience inappropriate hemodynamic conditions for their cell type: veins exposed to arterial flow, or arterial regions where flow becomes disturbed.
Clinical Implications: Rethinking Vascular Disease
Understanding why arteries get atherosclerosis while veins don't has important clinical implications. It explains why interventions targeting systemic risk factors—while crucial—cannot completely prevent disease. Even with optimal cholesterol levels, blood pressure, and glucose control, atherosclerosis will still develop preferentially at hemodynamically vulnerable sites.
It also suggests new therapeutic strategies. Rather than focusing solely on systemic risk reduction, we might target the local vascular environment. This could include:
- Designing better vein grafts that resist the pathological transformation when placed in arterial circulation
- Developing interventions that modify local hemodynamics to reduce disturbed flow patterns
- Creating therapies that enhance endothelial mechanosensing to maintain protective responses even under suboptimal flow conditions
The vein graft experience also highlights the importance of arterial conduits for bypass surgery. Internal mammary arteries, which are already adapted to arterial pressures and flow, show dramatically better long-term patency than vein grafts, precisely because they're already programmed for the arterial environment.
Veins Perfect Storm Of Protection
Veins don't develop atherosclerosis because of a perfect storm of protective factors: low-pressure hemodynamics that avoid the turbulent flow patterns that trigger inflammation, intrinsic cellular programming established during development that makes venous cells less responsive to atherogenic stimuli, and smooth muscle cells that don't exhibit the proliferative behavior necessary for plaque formation.
Understanding these differences isn't just academically interesting—it has practical implications for designing better vascular grafts, developing targeted therapies, and understanding why certain interventions work in arteries but not in veins, or vice versa.
The next time you look at an anatomy diagram showing the red arteries and blue veins, remember: they're not just different colors on a chart. They're fundamentally different organs, built from different molecular blueprints, operating under different physical laws, and destined for different diseases.
So Why Do Arteries Get ASCVD And Not Veins? It's Retention!
Arteries can retain LDL particles. Veins cannot. It’s that simple. See the LDL Particle Retention article.
As Tabas and Williams have proven in multiple studies, it's the retention, stupid. Event if you delete everything you read above, arteries have the ability to retain lipoproteins, veins do not.
As discussed above extensively, when veins are removed from the venous side and placed into the arterial side, they can “arterialize” and can eventually develop ASCVD. This happens when a patient undergoes coronary artery bypass surgery (CABG). Veins are used to bypass plugged up arteries and they will arterialize and can later develop ASCVD and need stents or repeat CABG. The only way to prevent this is to lower LDL-C to target levels, likely below 55 or 40 mg/dL in these patients.
The veins are placed in a higher pressure system when they are used for bypass.
Articles discussing why arteries get ASCVD and not veins:
Response to Retention:
https://www.ahajournals.org/doi/10.1161/01.ATV.15.5.551
More up to date information in Boren article:
https://academic.oup.com/eurheartj/article/41/24/2313/5735221
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7308544/
https://journals.lww.com/co-lipidology/fulltext/2016/10000/the_central_role_of_arterial_retention_of.6.aspx
We may not fully understand the chemical signaling pathways and mechanisms, but arteries retain LDL particles and veins do not, as LDL particles do not enter veins.
Conclusion: Environment Shapes Destiny
The question of why arteries develop atherosclerosis while veins don't reveals a fundamental principle in vascular biology: local mechanical environment shapes disease susceptibility as powerfully as systemic risk factors. Arteries develop plaques not simply because they're arteries, but because they experience hemodynamic conditions, particularly low and oscillatory shear stress at branch points, that promote endothelial dysfunction and LDL infiltration.
Veins remain protected not because they're inherently superior, but because their low-pressure, low-flow environment doesn't trigger the same pathological cascades. When veins are transplanted into arterial circulation, they rapidly develop the same disease, demonstrating that environment, not just vessel identity, drives atherosclerosis.
This understanding shifts how we think about cardiovascular disease prevention and treatment. While managing cholesterol, blood pressure, and other systemic risk factors remains essential, we must also consider the local vascular environment and the mechanical forces that ultimately determine where disease develops.
References
- Hemodynamic Shear Stress and Its Role in Atherosclerosis. Malek AM, Alper SL, Izumo S. JAMA. 1999;282(21):2035-42. doi:10.1001/jama.282.21.2035.
- Shear Stress, Arterial Identity and Atherosclerosis. Lehoux S, Jones EA. Thrombosis and Haemostasis. 2016;115(3):467-73. doi:10.1160/TH15-10-0791.
- Differences in Vascular Bed Disease Susceptibility Reflect Differences in Gene Expression Response to Atherogenic Stimuli. Deng DX, Tsalenko A, Vailaya A, et al. Circulation Research. 2006;98(2):200-8. doi:10.1161/01.RES.0000200738.50997.f2.
- Vascular Smooth Muscle Cells Derived From Atherosclerotic Human Arteries Exhibit Greater Adhesion, Migration, and Proliferation Than Venous Cells. Faries PL, Rohan DI, Wyers MC, et al. The Journal of Surgical Research. 2002;104(1):22-8. doi:10.1006/jsre.2002.6399.
- Hemodynamic Shear Stress and Its Role in Atherosclerosis. Malek AM, Alper SL, Izumo S. JAMA. 1999;282(21):2035-42. doi:10.1001/jama.282.21.2035.
- Shear Stress, Arterial Identity and Atherosclerosis. Lehoux S, Jones EA. Thrombosis and Haemostasis. 2016;115(3):467-73. doi:10.1160/TH15-10-0791.
- Differences in Vascular Bed Disease Susceptibility Reflect Differences in Gene Expression Response to Atherogenic Stimuli. Deng DX, Tsalenko A, Vailaya A, et al. Circulation Research. 2006;98(2):200-8. doi:10.1161/01.RES.0000200738.50997.f2.
- Vascular Smooth Muscle Cells Derived From Atherosclerotic Human Arteries Exhibit Greater Adhesion, Migration, and Proliferation Than Venous Cells. Faries PL, Rohan DI, Wyers MC, et al. The Journal of Surgical Research. 2002;104(1):22-8. doi:10.1006/jsre.2002.6399.
- Effect of Evolocumab on Saphenous Vein Graft Patency After Coronary Artery Bypass Surgery (NEWTON-CABG CardioLink-5): An International, Randomised, Double-Blind, Placebo-Controlled Trial. Verma S, Leiter LA, Teoh H, et al. Lancet (London, England). 2025;406(10509):1223-1234. doi:10.1016/S0140-6736(25)01633-2.
- External Support for Saphenous Vein Grafts in Coronary Artery Bypass Surgery: A Randomized Clinical Trial. Goldstein DJ, Puskas JD, Alexander JH, et al. JAMA Cardiology. 2022;7(8):808-816. doi:10.1001/jamacardio.2022.1437.
- Activation and Inflammation of the Venous Endothelium in Vein Graft Disease. Ward AO, Caputo M, Angelini GD, George SJ, Zakkar M. Atherosclerosis. 2017;265:266-274. doi:10.1016/j.atherosclerosis.2017.08.023.
- Aortocoronary Saphenous Vein Graft Disease: Pathogenesis, Predisposition, and Prevention. Motwani JG, Topol EJ. Circulation. 1998;97(9):916-31. doi:10.1161/01.cir.97.9.916.
- Saphenous Vein Graft Failure After Coronary Artery Bypass Surgery: Pathophysiology, Management, and Future Directions. Harskamp RE, Lopes RD, Baisden CE, de Winter RJ, Alexander JH. Annals of Surgery. 2013;257(5):824-33. doi:10.1097/SLA.0b013e318288c38d.
- Role of Endothelial Shear Stress in the Natural History of Coronary Atherosclerosis and Vascular Remodeling: Molecular, Cellular, and Vascular Behavior. Chatzizisis YS, Coskun AU, Jonas M, et al. Journal of the American College of Cardiology. 2007;49(25):2379-93. doi:10.1016/j.jacc.2007.02.059.
Still Have Questions? Stop Googling and Ask Dr. Alo.
You’ve read the science, but applying it to your own life can be confusing. I created the Dr. Alo VIP Private Community to be a sanctuary away from social media noise.
Inside, you get:
-
Direct Access: I answer member questions personally 24/7/365.
-
Weekly Live Streams: Deep dives into your specific health challenges.
-
Vetted Science: No fads, just evidence-based cardiology and weight loss.
Don't leave your heart health to chance. Get the guidance you deserve. All this for less than 0.01% the cost of health insurance! You can cancel at anytime!
[👉 Join the Dr. Alo VIP Community Today]




















