Are Non Steroidal Mineralocorticoid Antagonists Superior in Heart Failure! New Data!
Apr 18, 2026
New Non Steroidal Mineralocorticoid Receptor Antagonists In Heart Failure
There is a class of medications with decades of evidence behind them, a Class I guideline recommendation for heart failure, and mortality data that most drugs would envy. And yet they are prescribed to fewer than one in three patients who qualify for them. They are discontinued by nearly half of patients within 12 months of starting.
The drugs are mineralocorticoid receptor antagonists (MRAs), and the story of why they are underused tells you a lot about the gap between evidence and practice in cardiology. A comprehensive state-of-the-art review published in JACC: Heart Failure in December 2024 breaks down where the evidence stands and where the field is heading. Let me give you the full picture.
Two Type Of Heart Failure
There are two types of heart failure that both produce a lack of forward flow in the heart. There is systolic heart failure and diastolic heart failure. See my other blog posts on heart failure types and treatments. Today we will focus on a medication class that can be used in both.
Mineralocorticoid receptor antagonists are certainly indicated in systolic heart failure, but there role hasn't been defined clearly in diastolic heart failure or heart failure with preserved or mildly reduced ejection fraction. These are often abbreviated as HFpEF and HFmrEF.
In HFpEF and HFmrEF, it appears that standard MRAs have no effect, while the newer non steroidal MRAs have shown benefit.
Key Takeaways
|
MRAs are guideline-recommended for HFrEF with a Class I, Level A designation, yet fewer than 33% of eligible patients receive them. |
|
Hyperkalemia and sexual side effects (gynecomastia, impotence) are the main reasons for underuse and discontinuation. |
|
RALES and EMPHASIS-HF established a 30-37% reduction in cardiovascular death or HF hospitalization for steroidal MRAs in HFrEF. |
|
TOPCAT failed to show a significant benefit for spironolactone in HFpEF overall. SPIRIT-HF (ACC 2026) now adds another neutral result, though the trial enrolled only half its planned patients. |
|
Nonsteroidal MRAs (finerenone) are pulling ahead: FINEARTS-HF was positive, and the FDA approved finerenone for HFmrEF and HFpEF in July 2025. |
|
SPIRIT-HF numerically favored placebo over spironolactone, reinforcing that steroidal MRAs in HFpEF remain unproven and nonsteroidal agents appear to be the better path forward. |
|
SGLT2 inhibitors and potassium binders can mitigate hyperkalemia risk, removing a major barrier to MRA use in HFrEF. |
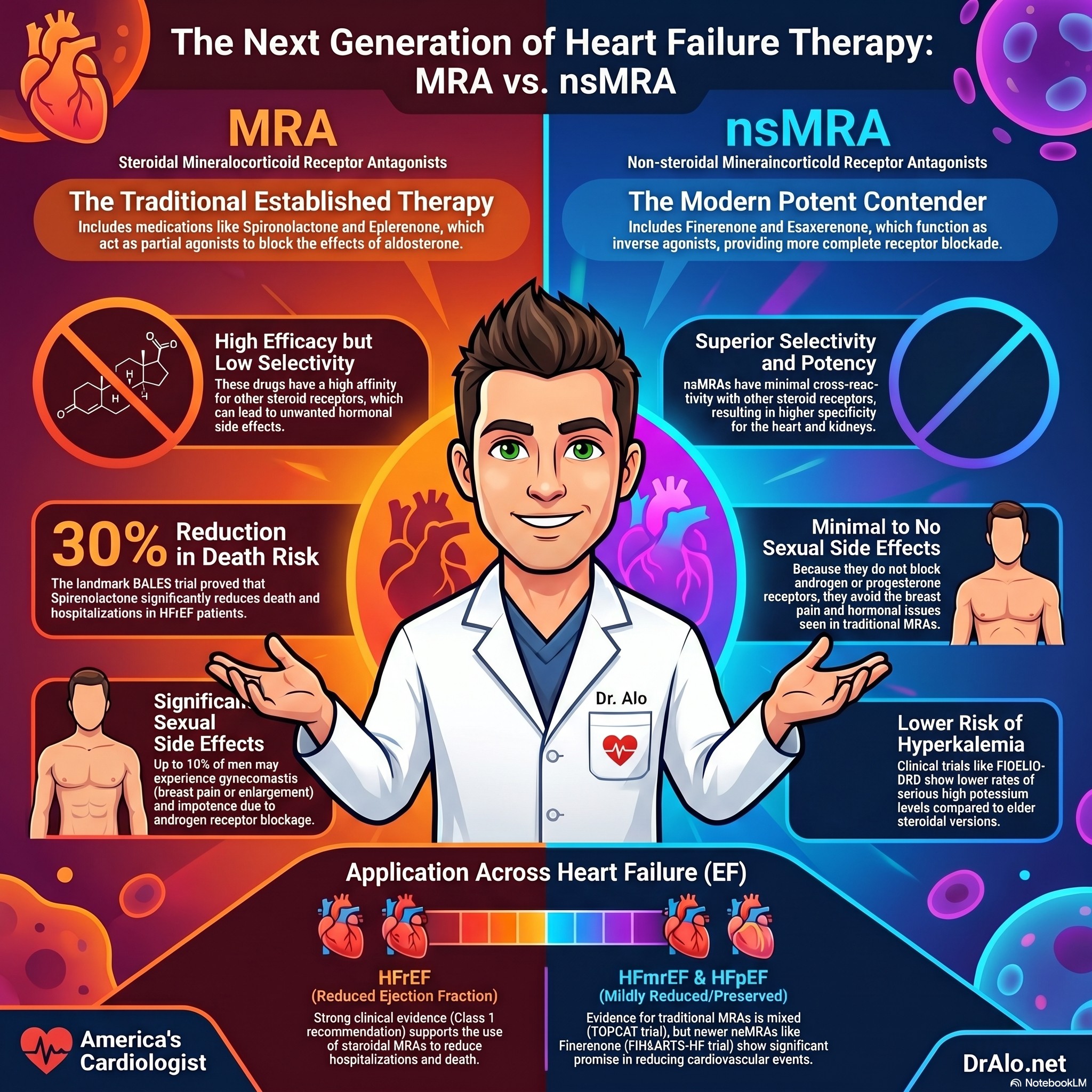
MRA vs nsMRA in Heart Failure Infographic Summary:
What Is a Mineralocorticoid Receptor Antagonist?
To understand why MRAs matter, you have to understand aldosterone. Aldosterone is a hormone made by your adrenal glands, and in heart failure, it gets overactivated. When that happens, several bad things follow: your kidneys retain sodium and water (worsening congestion), your heart develops fibrosis and pathological remodeling, and your blood vessels develop endothelial dysfunction.
MRAs directly block the mineralocorticoid receptor, preventing aldosterone from causing these downstream effects. The two steroidal MRAs in common use are spironolactone and eplerenone. Both have been around for decades and both have robust trial data. The problem is that by also binding to androgen and progesterone receptors, spironolactone in particular causes side effects that push patients and physicians away from it.
The Evidence in HFrEF: As Strong as It Gets
Three landmark trials established the case for MRAs in heart failure with reduced ejection fraction (HFrEF):
RALES (1999): Spironolactone 25 mg versus placebo in 1,663 patients with NYHA class III-IV HF and ejection fraction 35% or below. The result was a 30% reduction in the risk of death from any cause, plus significant reductions in cardiac hospitalizations and improvements in NYHA class. Severe hyperkalemia occurred in 2% of patients, which at the time was considered manageable.
EMPHASIS-HF (2011): Eplerenone versus placebo in 2,737 patients with NYHA class II HF and ejection fraction 30% or below. The composite of cardiovascular death or HF hospitalization was reduced by 37%. This trial was important because it proved benefit even in patients with milder symptoms, which expanded the eligible population considerably.
EPHESUS: Eplerenone in post-MI HF patients, showing significant reductions in cardiovascular death and hospitalization. The data are consistent across all three: MRAs save lives in HFrEF.
Based on this, both the ACC/AHA/HFSA and the ESC guidelines give MRAs a Class I, Level A recommendation for HFrEF. That is the highest possible designation. And still, usage sits around 33% in the United States and even lower in some international registries. In Japan, only 5.1% of patients reach the target dose.
So Why Are MRAs So Underused?
The RALES trial publication, while a landmark in cardiology, had an unintended consequence: a population-based time-series analysis showed that hospital admissions for hyperkalemia in HF patients on spironolactone plus ACE inhibitors tripled after RALES was published. The rate of hyperkalemia-associated hospitalization rose to 11 per 1,000 patients. Physicians noticed. Prescribing became cautious, then hesitant, and in many cases stopped.
The reasons for underuse cluster into a few categories:
- Hyperkalemia risk, particularly when combined with ACE inhibitors, ARBs, or in patients with kidney disease
- Gynecomastia and breast pain (10% of men in RALES reported this with spironolactone)
- Sexual side effects including impotence and menstrual irregularities
- Inadequate monitoring of potassium levels after initiation
- Clinician uncertainty about late initiation in long-term HFrEF patients
What the review highlights, and what I want every clinician reading this to know, is that the hyperkalemia concern is now much more manageable than it was in 1999. SGLT2 inhibitors have been shown to reduce the odds of developing hyperkalemia. Sacubitril-valsartan also mitigates the risk. And potassium-binding agents like patiromer have been shown to reduce hyperkalemia rates when used alongside MRAs. The clinical landscape has changed. The hesitancy should change with it.
MRAs in HFpEF: The Complicated Story
Heart failure with preserved ejection fraction (HFpEF, LVEF 50% or above) has been a harder nut to crack for MRAs. The pivotal trial was TOPCAT.
TOPCAT enrolled 3,445 patients with symptomatic HF and LVEF 45% or above, randomized to spironolactone versus placebo. The overall result was disappointing: the primary composite of cardiovascular death, aborted cardiac arrest, or HF hospitalization was not significantly reduced (HR: 0.89, P=0.14). Spironolactone did reduce HF hospitalizations modestly (HR: 0.83, P=0.04), but that was not enough to change guidelines substantially.
However, TOPCAT has a significant asterisk. When the data were analyzed by geographic region, a striking divergence emerged. Patients from the Americas showed meaningful benefit from spironolactone (HR: 0.82). Patients from Russia and Georgia showed none (HR: 1.10). Post-hoc analyses found that 30% of participants from Russia had undetectable levels of canrenone, the active metabolite of spironolactone, compared to only 3% from the United States and Canada. The evidence strongly suggests that a significant portion of the Russian/Georgian cohort was not actually taking the drug. In other words, TOPCAT may have been contaminated by non-adherence in the control-like regions, diluting what might otherwise have been a real signal.
Additionally, a secondary analysis of TOPCAT found that women derived significantly greater benefit from spironolactone than men with regard to all-cause mortality (HR: 0.66, P=0.01), with a significant interaction between sex and treatment arm. This is a clinically meaningful subgroup finding.
The Aldo-DHF trial studied spironolactone in a European HFpEF cohort and found improvements in diastolic function (E/e' ratio) but no change in peak VO2 or clinical symptoms. So spironolactone does appear to affect the underlying cardiac mechanics in HFpEF, but translating that into hard outcomes has been elusive with steroidal agents.
SPIRIT-HF (ACC 2026): The Latest Nail in Spironolactone's HFpEF Coffin
Fresh off the ACC 2026 Scientific Session in New Orleans, SPIRIT-HF is the most current attempt to settle the steroidal MRA question in HFpEF and HFmrEF. The trial enrolled patients 50 years and older with NYHA class II-IV HF, LVEF 40% or above, echocardiographic evidence of structural or functional cardiac abnormalities, and either a recent HF hospitalization or elevated NT-proBNP. Participants were randomized to spironolactone 25 mg (titrated to 50 mg) or placebo.
The results were not what anyone hoped for. Over two years, the rate of the primary composite endpoint of cardiovascular death or total HF hospitalizations per 100 patient-years was 10.9 in the spironolactone group versus 8.2 in the placebo group. That is numerically worse with the drug, not better (rate ratio 1.32; 95% CI 0.79-2.21), though the result was not statistically significant.
The trial lead investigator, Dr. Frank Edelmann, was candid: SPIRIT-HF is an underpowered trial. The study planned to enroll 1,564 patients but ultimately randomized only 730, roughly 47% of target, after the COVID-19 pandemic decimated funding and recruitment. With a 65% likelihood of a type II error, the trial cannot provide a definitive verdict. But what it can do is add to a pattern.
The picture from the data is telling even in its ambiguity. Half of spironolactone-treated patients had discontinued or near-zero drug intake by 21 months, compared to 31 months in the placebo group. The culprits were predictable: hypotension, hyperkalemia, and worsening renal function, with rate ratios ranging from 1.75 to 3.49 for these adverse events in the spironolactone arm.
An on-treatment analysis (censoring patients at the time of discontinuation) produced a more favorable rate ratio for spironolactone (0.94), and a prespecified meta-analysis combining SPIRIT-HF with the TOPCAT Americas cohort did show a signal of benefit. So the hypothesis that spironolactone might help in HFpEF patients who can tolerate it is not dead, but it remains unconfirmed.
The discussant at ACC 2026, Dr. Anu Lala, framed it precisely: SPIRIT-HF does not represent a failure of steroidal MRAs in this population because it is inconclusive. But Dr. Biykem Bozkurt added the key caveat that the critical question right now is whether there is a difference between steroidal and nonsteroidal MRAs in HFpEF, and based on the totality of evidence, the nonsteroidal agents are the ones with the positive trial results.
When you line up the scoreboard, the contrast is stark. TOPCAT: neutral overall, with adherence questions in a major geographic cohort. SPIRIT-HF: numerically trending toward harm, underpowered, discontinued at high rates. FINEARTS-HF (finerenone): met its primary endpoint. The FDA approved finerenone specifically for HFmrEF and HFpEF in July 2025. That is the direction the evidence is pointing, and clinicians should be reading it clearly.
Steroidal vs. Nonsteroidal MRAs: A Side-by-Side Look
The limitations of spironolactone and eplerenone drove the development of a new generation of MRAs that are chemically distinct and pharmacologically superior in several ways.
|
Feature |
Steroidal MRAs (Spiro / Eplerenone) |
Nonsteroidal MRAs (Finerenone, etc.) |
|
Selectivity for MR |
Lower (cross-reacts with androgen/progesterone receptors) |
Higher (minimal off-target receptor activity) |
|
Potency |
Moderate |
Comparable to or exceeding spironolactone |
|
Receptor activity |
Partial agonists |
Inverse agonists or partial antagonists |
|
Hyperkalemia risk |
Dose-dependent increase in K+ |
Lower rates; less effect on urinary Na+/K+ ratio |
|
Sexual side effects |
Gynecomastia, impotence, menstrual changes |
Minimal to none |
|
Antifibrotic activity |
Moderate |
Greater inhibition of profibrotic gene expression |
|
FDA approval in HF |
Yes (spironolactone, eplerenone) |
Not yet (finerenone approved for CKD/T2DM) |
Finerenone: The Nonsteroidal MRA Leading the Charge
Finerenone is the most studied nonsteroidal MRA and the one closest to changing clinical practice in heart failure. It has greater specificity for the mineralocorticoid receptor than either spironolactone or eplerenone, negligible cross-reactivity with androgen or progesterone receptors, and functions as an inverse agonist, which results in greater inhibition of profibrotic gene expression compared to the partial agonism of the steroidal agents.
The foundation of finerenone's evidence base comes from kidney disease trials in patients with type 2 diabetes:
- FIDELIO-DKD: Finerenone reduced the composite kidney outcome by 18% (HR: 0.82) and cardiovascular events by 14% (HR: 0.86) in patients with CKD and type 2 diabetes. Serious hyperkalemia occurred in only 1.6% of the finerenone group, far lower than historical rates with steroidal MRAs.
- FIGARO-DKD: Finerenone reduced the primary cardiovascular composite (CV death, nonfatal MI, nonfatal stroke, HF hospitalization) by 13% (HR: 0.87). Again, very low rates of serious hyperkalemia (0.7%).
- FIDELITY (pooled analysis): Across 13,000 patients, finerenone reduced CV outcomes by 14% (HR: 0.86) and HF hospitalization specifically by 22% (HR: 0.78). These are the kinds of numbers that translate into meaningful clinical benefit.
Then came FINEARTS-HF: finerenone versus placebo in patients with HFmrEF or HFpEF (LVEF 40% or above). This trial met its primary endpoint, achieving a statistically significant reduction in the composite of cardiovascular death and total heart failure events, defined as hospitalizations or urgent HF visits. This is a pivotal result. It is the first nonsteroidal MRA to show a positive outcome in the preserved/mildly reduced EF population and positions finerenone as a meaningful addition to the HFpEF therapeutic arsenal alongside SGLT2 inhibitors.
Other Nonsteroidal MRAs in the Pipeline
The field is not limited to finerenone. The review highlights two other nonsteroidal agents worth knowing about:
Esaxerenone is approved for hypertension in Japan and has shown renoprotective effects in type 2 diabetes as well as improvements in left ventricular mass index and BNP levels in HFpEF patients. It has a longer half-life and higher bioavailability than spironolactone and a cleaner side effect profile.
Balcinrenone (AZD9977) is a selective partial MR agonist designed to separate the cardiorenal protective effects from the electrolyte effects. In the MIRACLE trial (phase 2b), balcinrenone plus dapagliflozin did not significantly reduce UACR compared to dapagliflozin alone at 12 weeks, which was disappointing. However, the larger BalanceD-HF phase 3 trial is ongoing and will evaluate balcinrenone plus dapagliflozin against dapagliflozin alone for hard outcomes in HF patients with impaired kidney function. Results are expected in 2027.
Ocedurenone (KBP-5074) is another highly selective nonsteroidal MRA with preclinical renoprotective data and early clinical evidence of blood pressure reduction in CKD patients with uncontrolled hypertension. It has not yet been studied in HF specifically.
Upcoming Trials That Will Reshape This Space
The review maps out a rich pipeline of ongoing trials that will define how MRAs are used across the ejection fraction spectrum over the next several years:
- CONFIRMATION-HF: Finerenone plus an SGLT2 inhibitor versus standard of care in hospitalized HF patients, across all ejection fractions. This trial will directly test whether the two drug classes work synergistically and whether SGLT2 inhibitors indeed buffer the hyperkalemia risk of MRAs in real-time clinical practice.
- FINALITY-HF: Finerenone versus placebo in HFrEF patients who are intolerant of or ineligible for steroidal MRAs. This is critical for the 30 to 60% of HFrEF patients who should be on an MRA but cannot tolerate the side effects of spironolactone or eplerenone.
- REDEFINE-HF: Finerenone versus placebo in patients hospitalized with HFmrEF or HFpEF (LVEF 40% or above, NT-proBNP 1,000 pg/mL or above). This will expand the FINEARTS-HF data into the acute decompensated setting.
- SPIRIT-HF: Spironolactone versus placebo in HFmrEF or HFpEF, targeting 1,300 patients. This will provide a more rigorous assessment of the steroidal MRA question in preserved EF than TOPCAT delivered.
- SPIRRIT: A registry-randomized trial of spironolactone plus usual care versus usual care alone in 3,500 HFpEF patients. The registry-based design will provide real-world applicability that traditional RCTs often lack.
What This Means in Clinical Practice
If you are a clinician treating heart failure, the bottom line from this body of evidence is increasingly actionable. For HFrEF, MRAs are underprescribed, and the barriers that kept them out of your prescribing habits in the past are largely surmountable today. Using an SGLT2 inhibitor simultaneously reduces hyperkalemia risk. Potassium binders are available if needed. Eplerenone avoids the gynecomastia problem. The side effect profile should no longer be the deciding factor for most patients.
For HFmrEF and HFpEF, the data have spoken with increasing clarity. Finerenone received FDA approval for this indication in July 2025, based on the FINEARTS-HF result. SPIRIT-HF presented at ACC 2026 added another disappointing chapter for spironolactone in this population, albeit in an underpowered trial. Taken together, the pattern strongly favors reaching for a nonsteroidal MRA when you want MR antagonism in the preserved EF population.
If you are a patient with heart failure and you are not on an MRA, it is worth asking your cardiologist why. The answer may be reasonable: your kidney function or potassium levels may make it unsafe at this time. But if the answer is simply that nobody got around to it, that is a gap worth closing. The mortality data in HFrEF are too good to ignore. And in HFpEF, finerenone is now an FDA-approved option with a positive trial result behind it.
nsMRA and HFpEF and HFmrEF Bottom Line
Mineralocorticoid receptor antagonists have been fighting against their own side effect reputation for a quarter century. In HFrEF, the evidence for benefit was established in the 1990s and confirmed repeatedly since. The utilization gap is a healthcare quality problem, not an evidence problem.
In HFpEF and HFmrEF, the verdict is now coming in and it is increasingly clear: steroidal MRAs like spironolactone do not appear to be the right tool for this population. TOPCAT was neutral with adherence concerns. SPIRIT-HF, the most recent attempt to answer this question, was underpowered but numerically trended in the wrong direction, with high rates of side-effect-driven discontinuation reinforcing the core limitation of these agents.
Meanwhile, finerenone has delivered a positive result in FINEARTS-HF and received FDA approval for HFmrEF and HFpEF in July 2025. The pharmacological advantages of nonsteroidal MRAs, greater selectivity, no sexual side effects, lower hyperkalemia risk, and greater antifibrotic potency, are translating into real-world clinical outcomes where spironolactone has repeatedly fallen short.
The field is not ready to say spironolactone is useless in HFpEF. The SPIRRIT trial is ongoing and may yet provide a more definitive answer. But the weight of evidence right now points in one direction: if you are going to use an MRA in a patient with preserved or mildly reduced ejection fraction, finerenone is the agent with the evidence behind it. For HFrEF, fix the prescribing gap. For HFpEF, choose your agent wisely.
References:
Chang J, Ambrosy AP, Vardeny O, Van Spall HGC, Mentz RJ, Sauer AJ. Mineralocorticoid Antagonism in Heart Failure: Established and Emerging Therapeutic Role. JACC Heart Fail. 2024;12(12):1979-1993. DOI: 10.1016/j.jchf.2024.08.007
Edelmann F. SPIRIT-HF: Spironolactone in the Treatment of Heart Failure. Presented at: ACC 2026. March 29, 2026. New Orleans, LA. Reported via TCTMD.
Still Have Questions? Stop Googling and Ask Dr. Alo.
You’ve read the science, but applying it to your own life can be confusing. I created the Dr. Alo VIP Private Community to be a sanctuary away from social media noise.
Inside, you get:
-
Direct Access: I answer member questions personally 24/7/365.
-
Weekly Live Streams: Deep dives into your specific health challenges.
-
Vetted Science: No fads, just evidence-based cardiology and weight loss.
Don't leave your heart health to chance. Get the guidance you deserve. All this for less than 0.01% the cost of health insurance! You can cancel at anytime!
[👉 Join the Dr. Alo VIP Community Today]