LDL Cholesterol Below 60: Can It Prevent, Stop, or Reverse Heart Disease?
Mar 29, 2026
The 60 mg/dL Threshold: What the Science Really Says About LDL Cholesterol and Plaque Formation
For decades, the medical community has debated what constitutes an "optimal" LDL cholesterol level. While current guidelines recommend targets below 100 mg/dL for most people and below 70 mg/dL for high-risk individuals, emerging evidence suggests that atherosclerotic plaque may begin forming at much lower levels than previously thought. A growing body of research points to a critical threshold around 60 mg/dL, below which atherosclerosis appears to be virtually absent, and above which plaque steadily accumulates.
This isn't just theoretical speculation. Multiple landmark studies using advanced imaging techniques have documented this phenomenon in thousands of patients, fundamentally challenging our understanding of what "normal" cholesterol really means.
Infographic Summary Of Why LDL Below 60 Prevents Heart Disease

The PESA Study: A Paradigm Shift in Understanding "Normal" LDL
The Progression of Early Subclinical Atherosclerosis (PESA) study represents one of the most comprehensive investigations into the relationship between LDL cholesterol and plaque formation in apparently healthy individuals. Researchers examined 4,184 asymptomatic middle-aged participants using advanced imaging techniques to detect atherosclerosis in multiple vascular beds—carotid arteries, aorta, iliofemoral arteries, and coronary arteries.
The findings were striking. Among participants with LDL-C levels below 60 mg/dL, researchers found essentially no evidence of atherosclerosis. This suggests that 60 mg/dL may represent a minimum threshold needed for atherosclerotic plaque to develop in the first place.
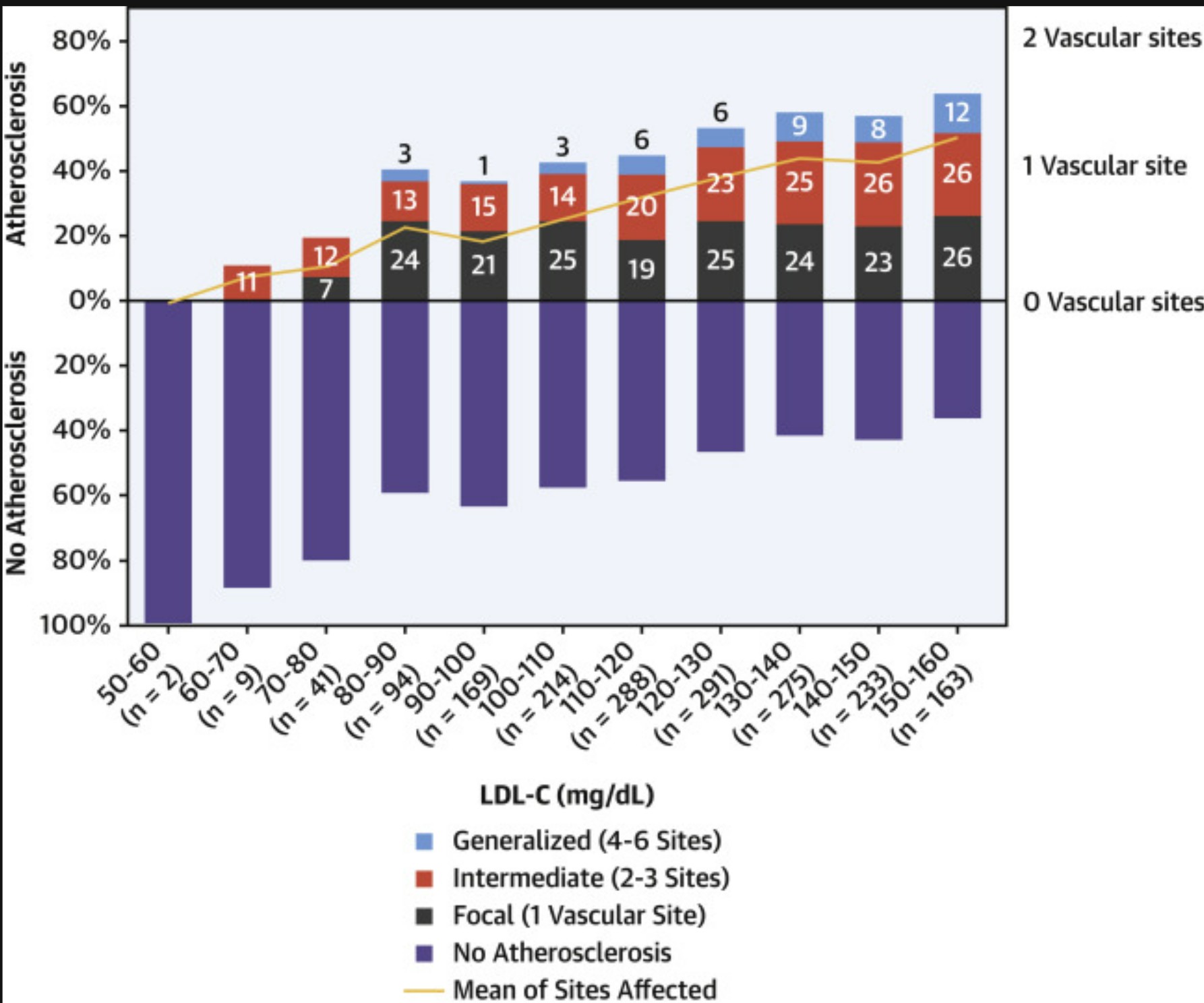
But perhaps even more revealing was what happened above this threshold. The study documented a linear, dose-dependent relationship between LDL-C levels and atherosclerosis prevalence across the entire spectrum of what we traditionally consider "normal" values. The prevalence of atherosclerosis increased progressively from just 11% in participants with LDL-C between 60-70 mg/dL to a remarkable 64% in those with levels between 150-160 mg/dL.
This wasn't just about whether plaque was present or absent. Higher LDL-C levels correlated with more extensive disease affecting multiple vascular territories. For every 10 mg/dL increase in LDL-C, participants had an 18% higher odds of having multiterritorial atherosclerosis, meaning plaque in multiple locations throughout their arterial system.
What makes these findings particularly important is that they occurred in people who would be considered low-risk by conventional standards. These were individuals without diabetes, without hypertension, without smoking history, and with LDL cholesterol levels that most physicians would consider acceptable or even optimal. Yet half of them already had subclinical atherosclerosis silently developing in their arteries.
LDL Below 60 mg/dL Prevents Heart Disease
In a 2018 paper where they looked at lifetime exposure to high LDL cholesterol, they found that lifelong exposure to high LDL cholesterol caused heart disease in a cumulative and dose response, linear pattern. The longer you were exposed to high LDL cholesterol, the more plaque burden. This is the paper that showed that those with 5000 mg years of LDL cholesterol exposure definitely had plaque.
One thing everyone missed from the article was that, at an LDL cholesterol below 60 mg/dL, there was no heart disease. Take a look at this graphic from the study.
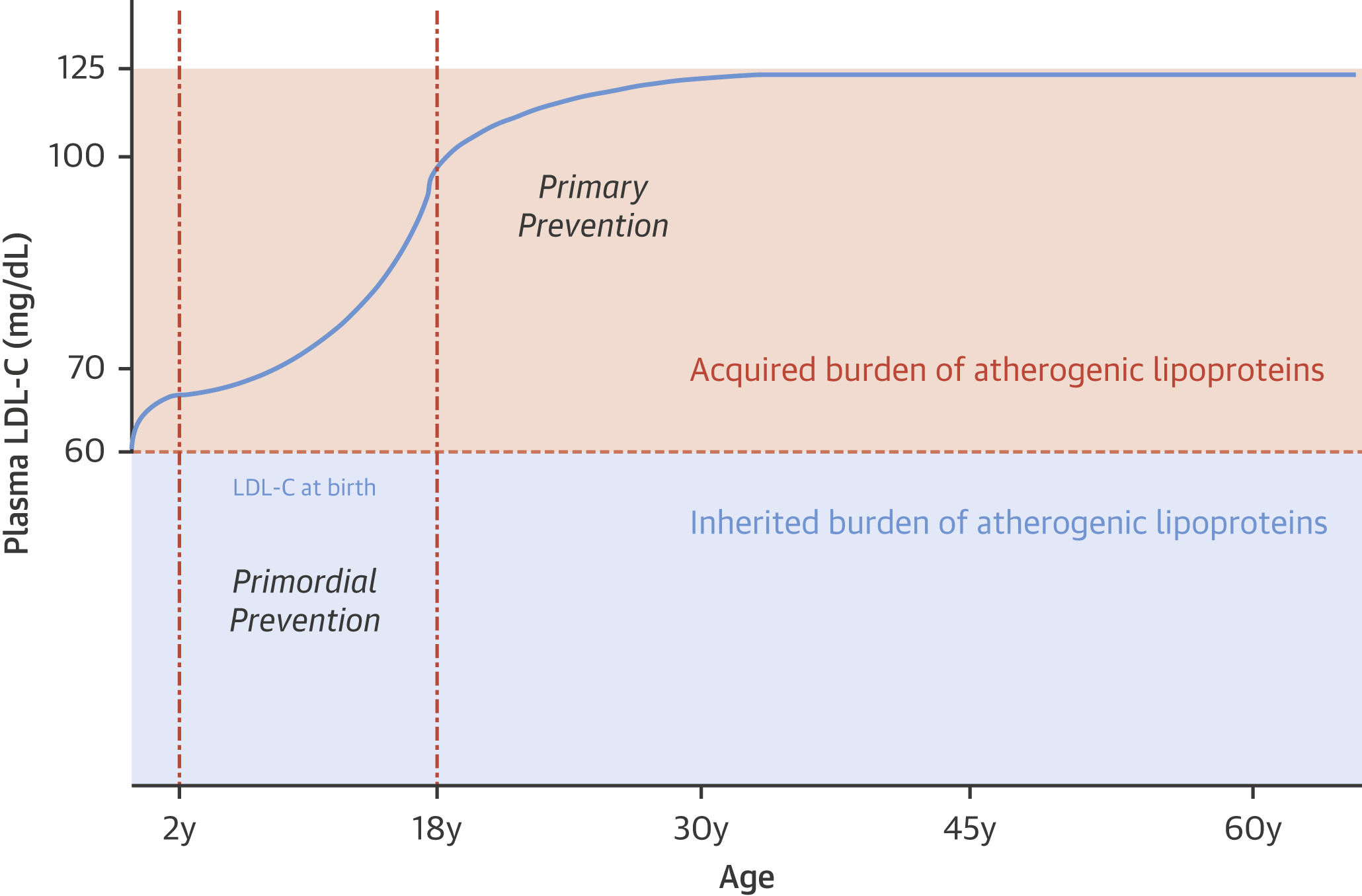
Notice that 60 mg/dL is the cut off for atherosclerotic heart disease. This means that individuals who have an LDL cholesterol below 60 mg/dL will not have heart disease. This has held true across many studies.
The Plaque Progression Studies: When Does Atherosclerosis Stop Growing?
While the PESA study revealed when plaque begins to form, a separate body of research using intravascular ultrasound (IVUS) and coronary angiography has examined a related but distinct question: at what LDL level does existing plaque stop progressing?
An integrated analysis of multiple IVUS studies demonstrated that plaque progression can be halted when LDL-C is lowered to 70-80 mg/dL. However, achieving actual plaque regression—shrinking existing atherosclerotic lesions—requires even lower levels. Analysis of invasive coronary angiography studies showed that coronary lesions do not progress when LDL-C is maintained below 70 mg/dL.
These findings align remarkably well with the PESA data. The threshold for preventing plaque formation (around 60 mg/dL) is very close to the threshold for halting plaque progression (70 mg/dL), suggesting a consistent biological mechanism across the spectrum from primary to secondary prevention.
FOURIER Trial (2017)
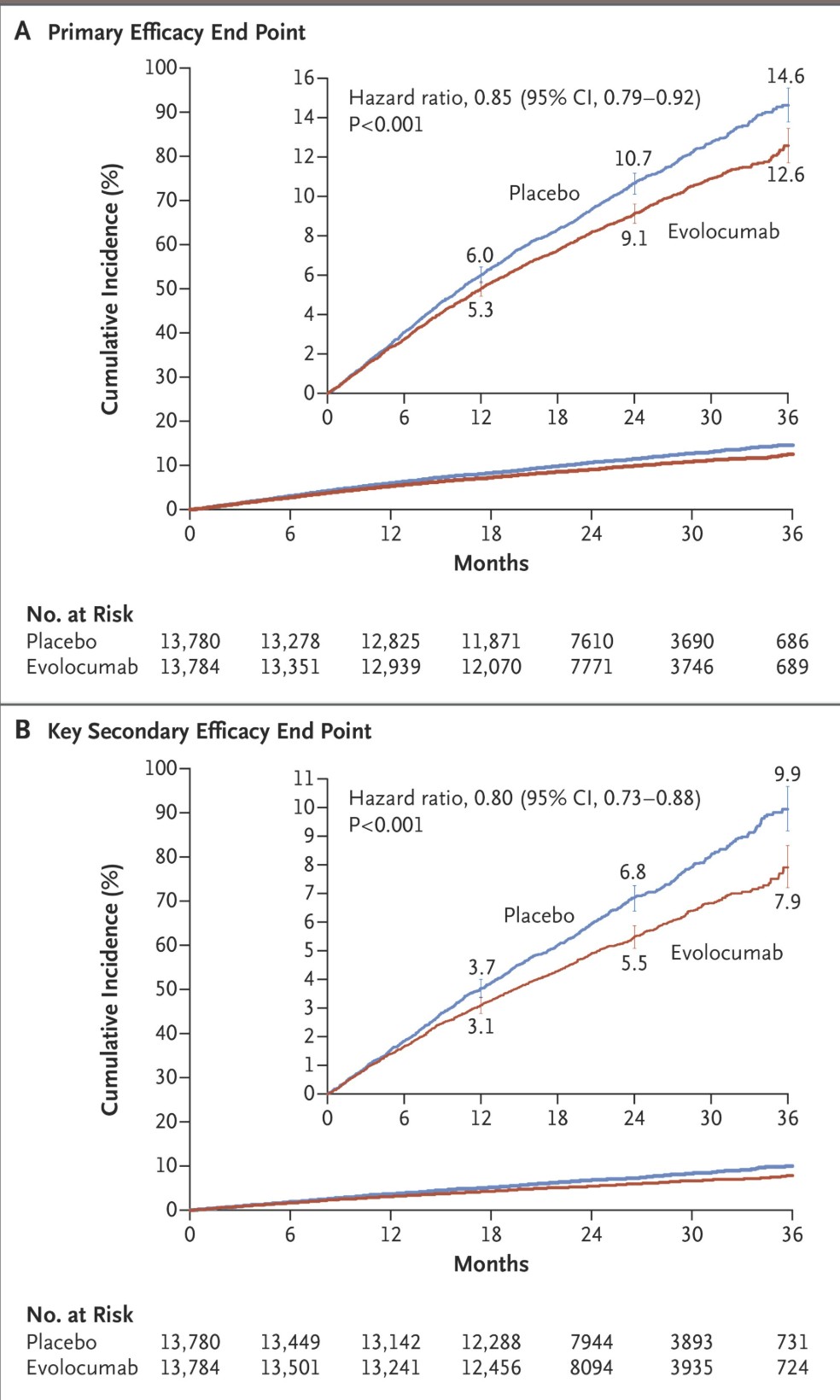
This landmark trial randomized 27,564 patients with established ASCVD to evolocumab (a PCSK9 inhibitor) versus placebo on top of statin therapy. The evolocumab group achieved a median LDL-C of 30 mg/dL, the lowest level recorded in a major cardiovascular outcomes trial. Over 2.2 years, evolocumab reduced major cardiovascular events by 15% (HR 0.85) and the key secondary endpoint of cardiovascular death, MI, or stroke by 20% (HR 0.80). Importantly, analyses showed a monotonic, nearly linear relationship between achieved LDL-C and cardiovascular risk continuing below 15 mg/dL, with no inflection point and no safety signal at extremely low levels.
GLAGOV: The Power of Intensive LDL Lowering
The GLAGOV trial provided dramatic evidence of what happens when LDL-C is reduced to very low levels in patients with established coronary disease. This study randomized 968 patients undergoing coronary angiography to receive either high-intensity statin therapy alone (achieving mean LDL-C of 93 mg/dL) or statin plus a PCSK9 inhibitor (achieving mean LDL-C of 37 mg/dL).
The results were unambiguous. The group achieving LDL-C of 37 mg/dL showed actual reduction in both percent atheroma volume and total atheroma volume—meaning their plaques were shrinking. This provided direct evidence that intensive LDL-lowering therapy can not only halt plaque progression but actually reverse existing atherosclerotic disease.
ASTEROID: Plaque Regression with Rosuvastatin
The ASTEROID trial examined 349 patients treated with high-dose rosuvastatin (40 mg daily) for 24 months, using IVUS to measure changes in coronary plaque burden. Treatment lowered LDL-C to a mean of 60.8 mg/dL, right at the critical threshold identified in PESA.
The results were encouraging: two-thirds of patients showed plaque regression, with a mean reduction in percent atheroma volume of 0.98%. This study demonstrated that achieving LDL-C levels around 60 mg/dL with statin therapy alone could produce measurable plaque regression in the majority of patients.
Serial Imaging Studies: The 70% Solution
A prospective computed tomography angiography (CTA) study evaluated the effect of different LDL targets on plaque progression. Patients treated to an LDL-C target below 70 mg/dL showed a 70% greater reduction in plaque progression compared to those with more modest LDL targets.
Additional studies using near-infrared spectroscopy demonstrated that the lipid core burden of coronary plaques, the dangerous cholesterol-rich centers that can rupture and cause heart attacks—decreased only after significant LDL reduction, not with lenient targets. This suggests that achieving very low LDL levels doesn't just slow plaque growth; it actually changes plaque composition in ways that make plaques more stable and less likely to cause acute events.
ODYSSEY OUTCOMES Trial
In nearly 19,000 patients with recent acute coronary syndrome, alirocumab achieved a mean LDL-C of 38 mg/dL and reduced the primary composite endpoint by 15% (absolute risk reduction 1.6 percentage points) over 2.8 years, with a trend toward reduced all-cause mortality.
JUPITER: Primary Prevention with Intensive Therapy
The JUPITER trial examined primary prevention in individuals without established cardiovascular disease but with elevated inflammatory markers. Participants randomized to high-intensity rosuvastatin therapy achieved greater LDL-C reduction and experienced significantly fewer cardiovascular events compared to placebo.
This trial helped establish that intensive LDL-lowering provides benefit even in primary prevention, supporting the concept that earlier and more aggressive intervention may prevent atherosclerosis from developing in the first place.
JUPITER Trial Subanalysis
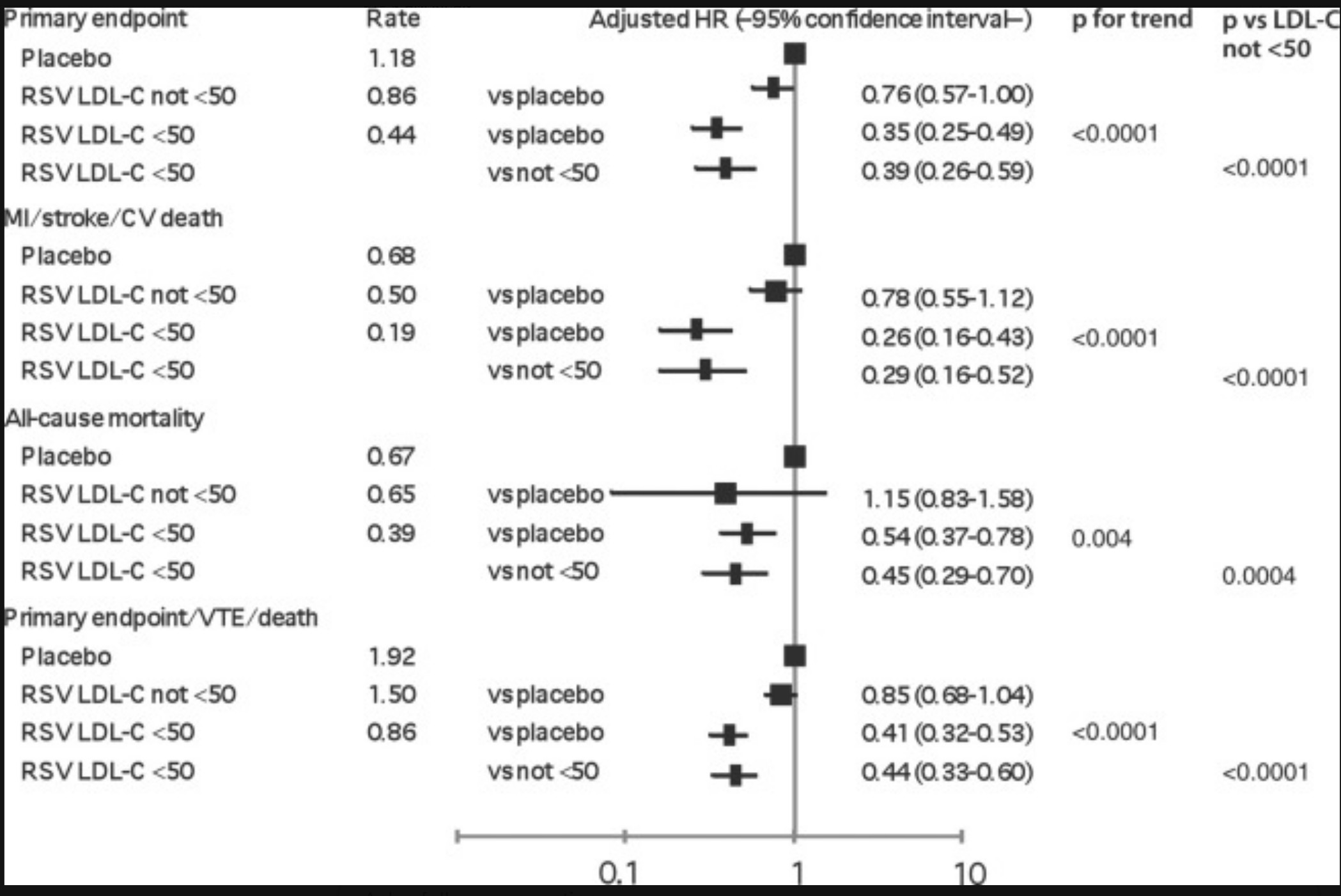
Among participants randomized to rosuvastatin, those achieving LDL-C below 50 mg/dL had a 65% reduction in the primary cardiovascular endpoint compared to placebo, versus only 24% reduction in those not reaching this threshold. There was also a 46% reduction in all-cause mortality among those achieving LDL-C below 50 mg/dL.
FOURIER OLE With Very Low LDL Levels
The FOURIER-OLE (Open-Label Extension) study provides the most robust long-term data on achieving very low LDL-C levels, including below 20 mg/dL. In this prespecified analysis of 6,559 patients followed for a median of 5 additional years (total follow-up up to 8.6 years), there was a monotonic relationship between lower achieved LDL-C levels, down to below 20 mg/dL, and lower cardiovascular risk, with no significant safety concerns.
Key Findings from FOURIER-OLE
Among the 6,559 patients with available LDL-C data:
-
24% (1,604 patients) achieved LDL-C below 20 mg/dL
-
40% achieved LDL-C 20 to <40 mg/dL
-
16% achieved LDL-C 40 to <55 mg/dL
-
7% achieved LDL-C 55 to <70 mg/dL
-
12% achieved LDL-C ≥70 mg/dL
There was a highly significant monotonic relationship between lower achieved LDL-C levels and reduced risk of both the primary endpoint (cardiovascular death, MI, stroke, hospitalization for unstable angina, or coronary revascularization) and the key secondary endpoint (cardiovascular death, MI, or stroke), with adjusted P-trend <0.0001 for each endpoint, extending down to levels below 20 mg/dL.
Safety at Ultra-Low LDL-C Levels
Critically, no statistically significant associations existed between lower achieved LDL-C levels and increased risk of safety outcomes, including:
-
Serious adverse events
-
New or recurrent cancer
-
Cataract-related adverse events
-
Hemorrhagic stroke
-
New-onset diabetes
-
Neurocognitive adverse events
-
Muscle-related events
-
Non-cardiovascular death
Original FOURIER Trial Data on Ultra-Low LDL-C
In the original FOURIER trial, an exploratory subgroup of 504 patients achieved LDL-C below 10 mg/dL (median 7 mg/dL, IQR 5-9 mg/dL). These patients had adjusted hazard ratios of 0.69 (95% CI 0.49-0.97) for the primary endpoint and 0.59 (95% CI 0.37-0.92) for the key secondary endpoint compared to patients with LDL-C ≥100 mg/dL, representing a 41% relative risk reduction for cardiovascular death, MI, or stroke. Neither serious adverse events nor adverse events leading to drug discontinuation occurred in excess in these ultra-low LDL-C groups.
The following figure from the FOURIER trial demonstrates the sustained LDL-C lowering and cardiovascular benefit with evolocumab:
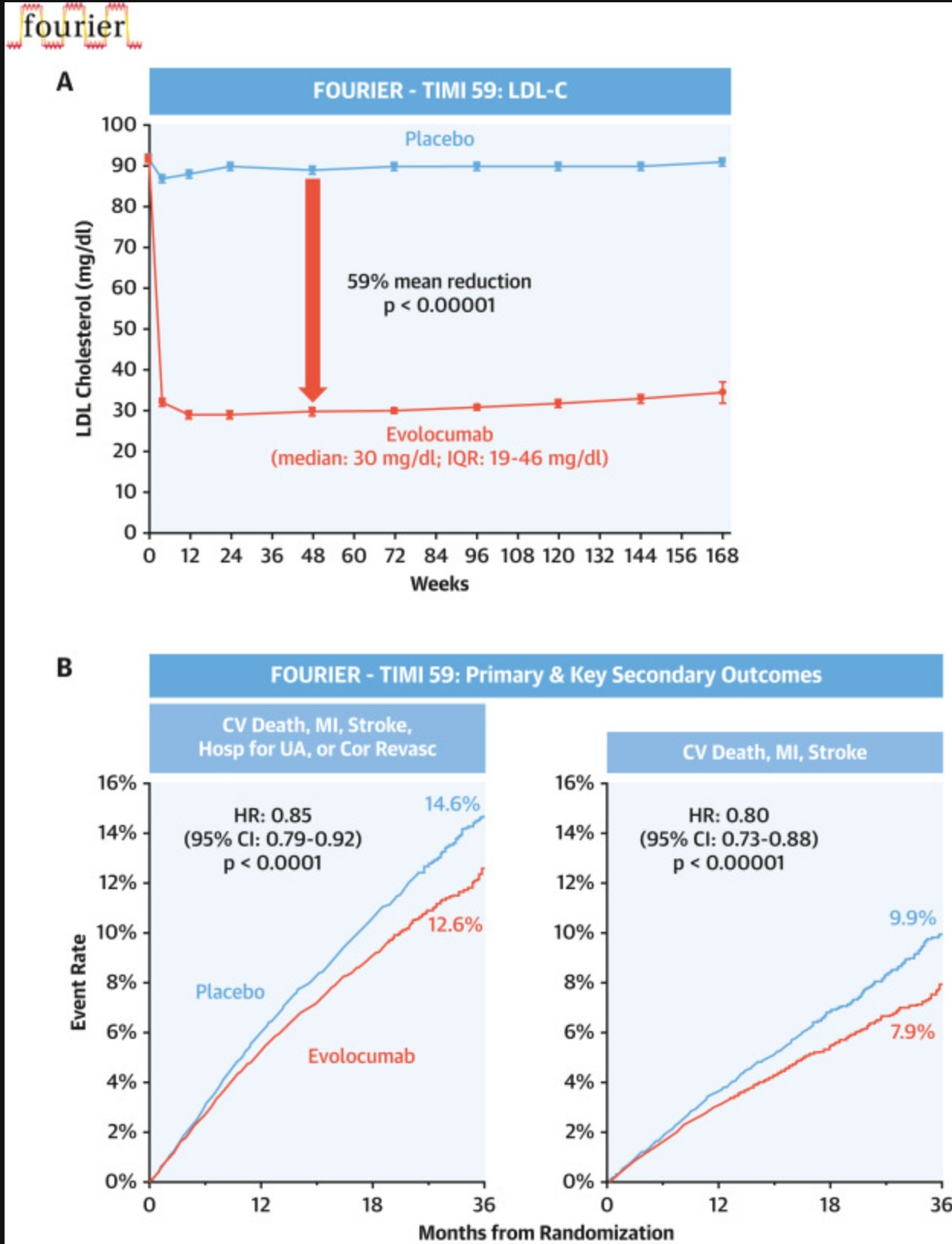
Meta-Analyses: The Big Picture
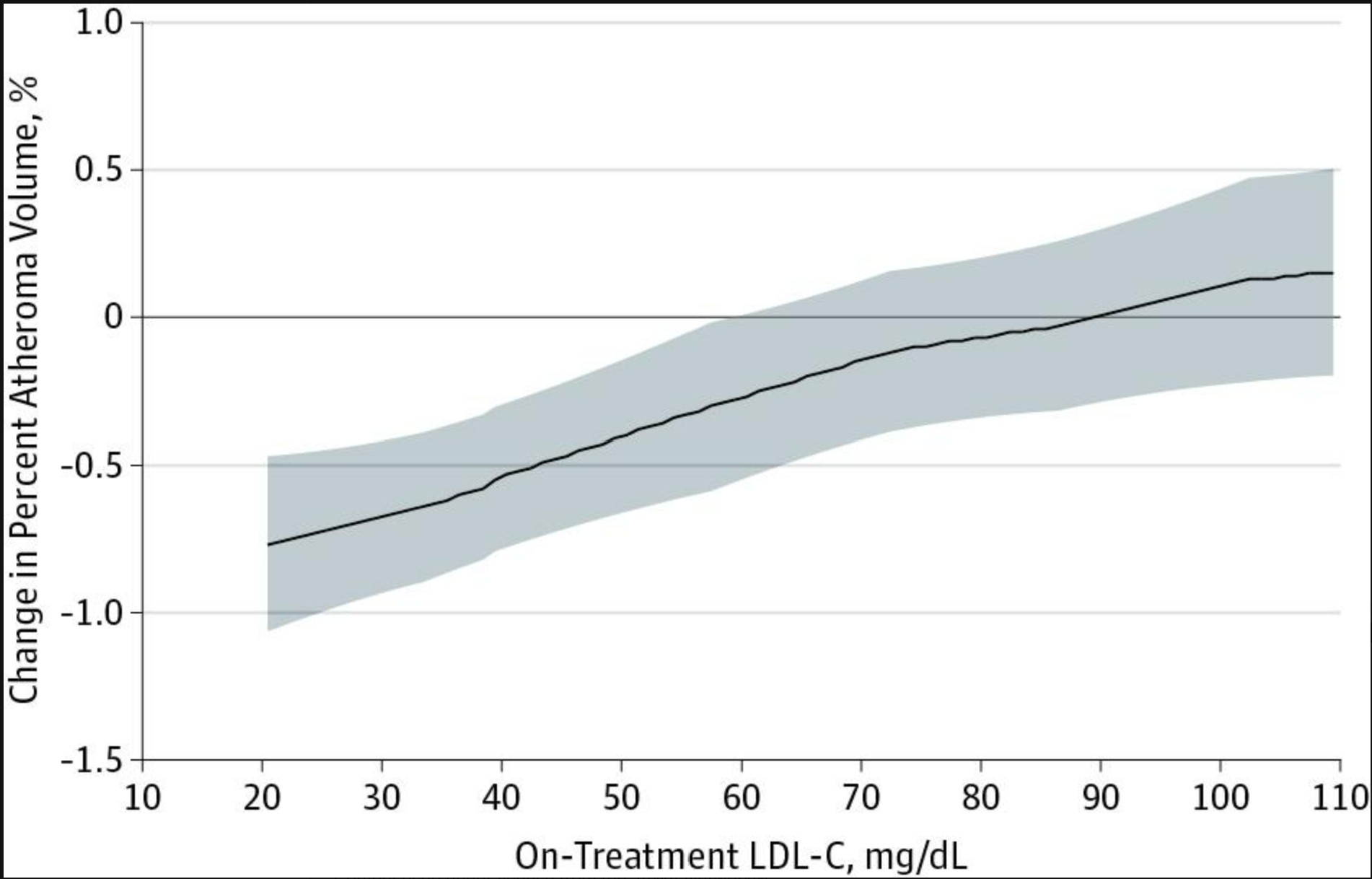
A comprehensive meta-analysis of 51 studies involving 9,113 patients examined the effects of various lipid-lowering therapies on plaque regression. The analysis confirmed that lipid-lowering treatments reduced both percent atheroma volume and total atheroma volume, with plaque regression primarily driven by high-intensity statin therapy achieving very low LDL-C levels.
Another meta-analysis of eight trials (1,759 patients) evaluated adding ezetimibe or PCSK9 inhibitors to statin therapy. The results showed that for every 10% decrease in LDL-C, there was approximately 1.0 mm³ regression in total atheroma volume. This dose-response relationship reinforces that when it comes to LDL-lowering and plaque regression, lower is indeed better.
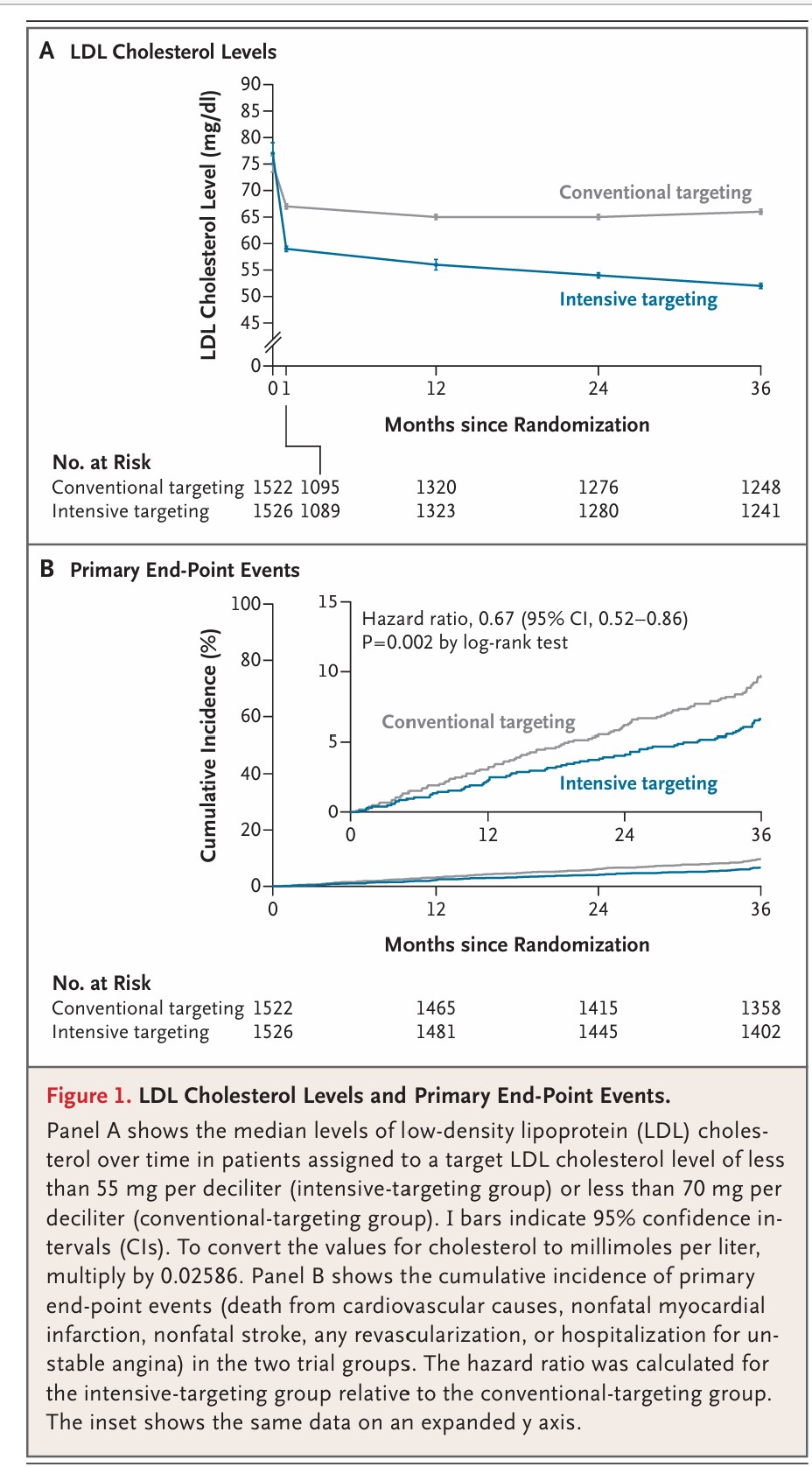
March 2026 Ez-PAVE Trial On Intensive Therapy: LDL 70 vs 55 New England Journal of Medicine
A new randomized trial in The New England Journal of Medicine helps answer one of the most important cholesterol questions in preventive cardiology: is aiming for an LDL below 55 mg/dL actually better than the traditional goal of below 70 mg/dL? In the Ez-PAVE trial, investigators enrolled 3,048 patients with established atherosclerotic cardiovascular disease and assigned them to one of those two LDL targets. Over a median follow-up of 3 years, the lower-target group achieved a median LDL of 56 mg/dL, compared with 66 mg/dL in the standard-target group. That difference translated into a meaningful reduction in major cardiovascular events.
Specifically, the composite rate of cardiovascular death, heart attack, stroke, revascularization, or hospitalization for unstable angina was 6.6% in the group targeting LDL below 55 mg/dL, compared with 9.7% in the group targeting below 70 mg/dL. That is a 33% relative risk reduction (hazard ratio 0.67, P=0.002). The benefit appeared to be driven largely by fewer nonfatal heart attacks and fewer repeat revascularization procedures, which is exactly what clinicians hope to prevent in high-risk patients. Just as important, there was no major safety penalty for aiming lower: rates of new-onset diabetes, worsening glycemic control, muscle symptoms, cancer, liver enzyme elevation, and CK elevation were similar between groups, while creatinine elevation was actually less common in the intensive-target group.
The big takeaway is that this was not simply another study showing that stronger cholesterol drugs work. It was a study testing the LDL target itself, and the lower target won. For patients who already have cardiovascular disease, 55 mg/dL should probably be viewed as the default goal rather than an optional “extra aggressive” target. The main caveat is that this trial was conducted in South Korea, was open-label, and included patients with existing ASCVD, so the strongest conclusion applies to secondary prevention, not necessarily to every person in the general population. Still, for people who have already had a cardiovascular event or clearly have atherosclerotic disease, this study makes a compelling case that lower is better, and 55 mg/dL is probably where we should be aiming.
What's more impressive is that every single subgroup did better, see below:
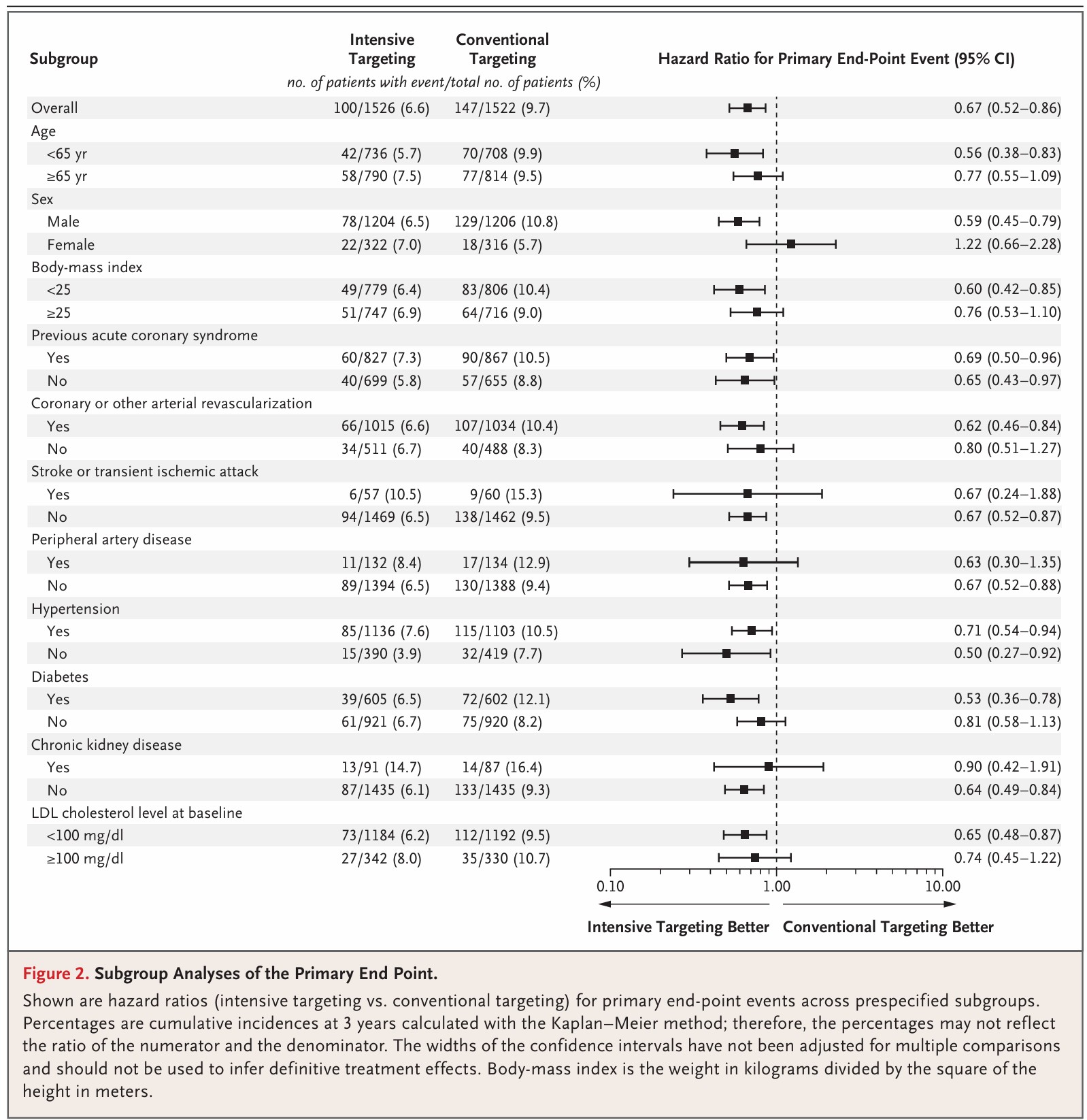
What All This Data Means for Clinical Practice
The 2026 ACC/AHA Dyslipidemia Guidelines have begun to incorporate these findings, now recommending treatment to an LDL-C goal below 70 mg/dL in high-risk primary prevention patients. The guidelines explicitly acknowledge that cumulative lifetime exposure to elevated atherogenic lipoproteins is closely associated with cardiovascular events.
Data from the CARDIA cohort demonstrated that even modest incremental increases in LDL-C over time were associated with significantly increased event rates over 16 years of follow-up. Similarly, the Framingham Heart Study Offspring cohort showed that cumulative exposure to elevated cholesterol in young adulthood increased subsequent coronary heart disease risk in a dose-dependent fashion.
These observations have profound implications. They suggest that:
1. The concept of "normal" LDL cholesterol may need revision. Values between 100-130 mg/dL are statistically normal in Western populations, but they're clearly not biologically optimal if atherosclerosis is silently progressing. Which is why I recommend much lower values.
2. Earlier intervention may be warranted. If plaque begins accumulating above 60 mg/dL and cumulative exposure matters, waiting until someone develops clinical disease or reaches a certain age may mean missing a critical window for prevention.
3. Treatment targets should be individualized based on risk. For someone with established disease or very high risk, achieving LDL-C levels of 50-60 mg/dL or even lower may be necessary to achieve plaque regression.
4. Imaging may help guide therapy. Detecting subclinical atherosclerosis in its early stages could identify individuals who would benefit from more intensive LDL-lowering, even if their traditional risk scores suggest otherwise.
The Biological Plausibility
Why would 60 mg/dL represent a threshold for atherosclerosis development? The answer likely relates to the balance between LDL infiltration into the arterial wall and the body's ability to remove it.
At very low LDL-C levels (below 60 mg/dL), the rate of LDL particle entry into the arterial wall may be slow enough that the body's natural clearance mechanisms can keep pace, preventing accumulation. Above this threshold, LDL infiltration exceeds clearance capacity, leading to progressive accumulation, oxidation, and the inflammatory cascade that characterizes atherosclerosis.
This is consistent with observations from populations with genetically low LDL cholesterol and from newborn humans, who typically have LDL-C levels around 30-70 mg/dL and show no evidence of atherosclerosis.
Unanswered Questions and Future Directions
While the evidence for a 60-70 mg/dL threshold is compelling, important questions remain:
- Would population-wide strategies to achieve LDL-C below 60 mg/dL from early adulthood prevent most cardiovascular disease?
- Are there long-term safety concerns with maintaining very low LDL levels for decades?
- How do we balance the benefits of intensive LDL-lowering against medication costs, side effects, and patient preferences?
- Can we identify which individuals are most likely to benefit from very aggressive LDL-lowering?
The upcoming SCOT-HEART 2 trial, which will compare treatment based on identification of subclinical atherosclerosis by coronary CTA with treatment based on standard risk factor scores, may help answer some of these questions.
Conclusion
The convergence of evidence from PESA, JUPITER, ASTEROID, GLAGOV, FOURIER, ODYESSEY, and numerous other trials paints a consistent picture: atherosclerotic plaque formation begins at LDL-C levels above approximately 60 mg/dL, plaque progression can be halted at levels below about 60-70 mg/dL, and plaque regression requires even lower levels, often below 50-60 mg/dL.
This represents a fundamental shift in how we think about cholesterol management. Rather than asking "Is this patient's LDL high enough to treat?" we might instead ask "Is this patient's LDL low enough to prevent disease?"
For clinicians, these findings support a more proactive approach to lipid management, particularly in patients with long life expectancy who will accumulate decades of exposure to atherogenic lipoproteins. For patients, it underscores the importance of knowing your numbers and understanding that "normal" on a lab report doesn't necessarily mean "optimal" for long-term cardiovascular health.
The science is clear: when it comes to LDL cholesterol and atherosclerosis, lower is better, and the threshold for disease development is much lower than most people realize.
References:
Subclinical Atherosclerosis & Plaque Threshold Studies
-
PESA (Progression of Early Subclinical Atherosclerosis) Study – Fernández-Friera L, Fuster V, López-Melgar B, et al. Normal LDL-Cholesterol Levels Are Associated With Subclinical Atherosclerosis in the Absence of Risk Factors. Journal of the American College of Cardiology. 2017;70(24):2979-2991. doi:10.1016/j.jacc.2017.10.024. https://pubmed.ncbi.nlm.nih.gov/29241485
-
PESA Study Overview – Ibanez B, Fernández-Ortiz A, Fernández-Friera L, et al. Progression of Early Subclinical Atherosclerosis (PESA) Study: JACC Focus Seminar 7/8. Journal of the American College of Cardiology. 2021;78(2):156-179. doi:10.1016/j.jacc.2021.05.011. https://pubmed.ncbi.nlm.nih.gov/34238438
Plaque Regression Trials
-
GLAGOV Trial – Nicholls SJ, Puri R, Anderson T, et al. Effect of Evolocumab on Progression of Coronary Disease in Statin-Treated Patients: The GLAGOV Randomized Clinical Trial. JAMA. 2016;316(22):2373-2384. doi:10.1001/jama.2016.16951. https://jamanetwork.com/journals/jama/fullarticle/2584184
-
ASTEROID Trial – Referenced in: Slipczuk L, Blankstein R, Bucciarelli-Ducci C, et al. State of the Art: Evaluation and Medical Management of Nonobstructive Coronary Artery Disease in Patients With Chest Pain: A Scientific Statement From the American Heart Association. Circulation. 2025;152(23):e443-e466. doi:10.1161/CIR.0000000000001394. https://www.ahajournals.org/doi/10.1161/CIR.0000000000001394
PCSK9 Inhibitor Cardiovascular Outcomes Trials
-
FOURIER Trial – Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and Clinical Outcomes in Patients With Cardiovascular Disease. New England Journal of Medicine. 2017;376(18):1713-1722. doi:10.1056/NEJMoa1615664. https://www.nejm.org/doi/full/10.1056/NEJMoa1615664
-
FOURIER Trial – Very Low LDL-C Subanalysis – Giugliano RP, Pedersen TR, Park JG, et al. Clinical Efficacy and Safety of Achieving Very Low LDL-cholesterol Concentrations With the PCSK9 Inhibitor Evolocumab: A Prespecified Secondary Analysis of the FOURIER Trial. Lancet. 2017;390(10106):1962-1971. doi:10.1016/S0140-6736(17)32290-0. https://pubmed.ncbi.nlm.nih.gov/28859947
-
FOURIER-OLE (Open-Label Extension) – Gaba P, O'Donoghue ML, Park JG, et al. Association Between Achieved Low-Density Lipoprotein Cholesterol Levels and Long-Term Cardiovascular and Safety Outcomes: An Analysis of FOURIER-OLE. Circulation. 2023;147(16):1192-1203. doi:10.1161/CIRCULATIONAHA.122.063399. https://pubmed.ncbi.nlm.nih.gov/36779348
-
FOURIER/FOURIER-OLE Stroke Analysis – Monguillon V, Kelly PJ, O'Donoghue ML, et al. Efficacy and Safety of Very Low Achieved LDL Cholesterol in Patients With Previous Ischemic Stroke. Circulation. 2026;153(2):86-93. doi:10.1161/CIRCULATIONAHA.125.077549. https://pubmed.ncbi.nlm.nih.gov/41178569
-
ODYSSEY OUTCOMES Trial – Referenced in: Michos ED, McEvoy JW, Blumenthal RS. Lipid Management for the Prevention of Atherosclerotic Cardiovascular Disease. New England Journal of Medicine. 2019;381(16):1557-1567. doi:10.1056/NEJMra1806939. https://www.nejm.org/doi/full/10.1056/NEJMra1806939
Statin Trials with Very Low LDL-C Outcomes
- JUPITER Trial – LDL <50 mg/dL Subanalysis – Hsia J, MacFadyen JG, Monyak J, Ridker PM. Cardiovascular Event Reduction and Adverse Events Among Subjects Attaining Low-Density Lipoprotein Cholesterol <50 mg/dL With Rosuvastatin: The JUPITER Trial. Journal of the American College of Cardiology. 2011;57(16):1666-75. doi:10.1016/j.jacc.2010.09.082. https://pubmed.ncbi.nlm.nih.gov/21492764
- Intensive LDL Cholesterol Targeting in Atherosclerotic Cardiovascular Disease https://www.nejm.org/doi/full/10.1056/NEJMoa2600283
- Impact of Lipids on Cardiovascular Health. The 5000mg Cholesterol Years. https://www.jacc.org/doi/pdf/10.1016/j.jacc.2018.06.046
Meta-Analyses
- Very Low LDL-C Meta-Analysis (8 Statin Trials) – Boekholdt SM, Hovingh GK, Mora S, et al. Very Low Levels of Atherogenic Lipoproteins and the Risk for Cardiovascular Events: A Meta-Analysis of Statin Trials. Journal of the American College of Cardiology. 2014;64(5):485-94. doi:10.1016/j.jacc.2014.02.615. https://pubmed.ncbi.nlm.nih.gov/25082583
Review Articles & State-of-the-Art Summaries
-
JACC State-of-the-Art Review on Subclinical Atherosclerosis – Ahmadi A, Argulian E, Leipsic J, Newby DE, Narula J. From Subclinical Atherosclerosis to Plaque Progression and Acute Coronary Events: JACC State-of-the-Art Review. Journal of the American College of Cardiology. 2019;74(12):1608-1617. doi:10.1016/j.jacc.2019.08.012. https://pubmed.ncbi.nlm.nih.gov/31537271
-
JACC Health Promotion Series on Lipids – Ference BA, Graham I, Tokgozoglu L, Catapano AL. Impact of Lipids on Cardiovascular Health: JACC Health Promotion Series. Journal of the American College of Cardiology. 2018;72(10):1141-1156. doi:10.1016/j.jacc.2018.06.046. https://pubmed.ncbi.nlm.nih.gov/30165986
-
TIMI Study Group Focus Seminar – Sabatine MS, Braunwald E. Thrombolysis in Myocardial Infarction (TIMI) Study Group: JACC Focus Seminar 2/8. Journal of the American College of Cardiology. 2021;77(22):2822-2845. doi:10.1016/j.jacc.2021.01.060. https://pubmed.ncbi.nlm.nih.gov/34082913
Clinical Practice Guidelines
- 2026 ACC/AHA Dyslipidemia Guidelines – Blumenthal RS, Morris PB, Gaudino M, et al. 2026 ACC/AHA/AACVPR/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Dyslipidemia. Journal of the American College of Cardiology. 2026. doi:10.1016/j.jacc.2025.11.016. https://linkinghub.elsevier.com/retrieve/pii/S0735-1097(25)10254-4
Still Have Questions? Stop Googling and Ask Dr. Alo.
You’ve read the science, but applying it to your own life can be confusing. I created the Dr. Alo VIP Private Community to be a sanctuary away from social media noise.
Inside, you get:
-
Direct Access: I answer member questions personally 24/7/365.
-
Weekly Live Streams: Deep dives into your specific health challenges.
-
Vetted Science: No fads, just evidence-based cardiology and weight loss.
Don't leave your heart health to chance. Get the guidance you deserve. All this for less than 0.01% the cost of health insurance! You can cancel at anytime!
[👉 Join the Dr. Alo VIP Community Today]




















