Fish Oil, Garlic, Red Yeast Rice, and Cinnamon Do Not Lower Your Cholesterol
Apr 08, 2026
Which Supplements Prevent Heart Disease?
The SPORT trial, published in the Journal of the American College of Cardiology in 2023, is the first rigorous randomized controlled trial to pit six of the most popular cholesterol supplements head-to-head against both placebo and a low-dose statin. The results are damning for the supplement industry and critically important for anyone managing their cardiovascular risk.
Let's talk about supplements that reduce heart disease. Or do they?
I want to talk about something I see constantly in my cardiology clinic. A patient comes in with an LDL of 145 mg/dL and a 10-year cardiovascular risk of 9%. They need a statin. I recommend one. And they tell me they are already taking something for their cholesterol.
Fish oil. Red yeast rice. Turmeric. Garlic. Plant sterols. Cinnamon. CoQ10.
What gives?
When I ask if they know whether it is working, the answer is almost always no. They heard it was good for the heart. Their friend takes it. They saw it at the pharmacy next to the statins. It said 'heart health' on the label. The price was right. And it felt like they were doing something.
This is one of the most consequential gaps in cardiovascular care. Patients with real, quantifiable, guideline-indicated risk are bypassing proven therapy in favor of supplements that have never been rigorously tested. And the supplement industry, operating under the extraordinarily permissive 1994 Dietary Supplement Health and Education Act, has been allowed to imply clinical benefit without ever having to prove it.
The SPORT trial, published in the Journal of the American College of Cardiology in January 2023, is the first properly designed, randomized, placebo-controlled clinical trial to test the six most commonly used cholesterol supplements directly against both a placebo and a proven medication simultaneously. The investigators, from the Cleveland Clinic and led by Dr. Steven Nissen, put the products that millions of Americans are buying in the belief that they are protecting their hearts to a formal scientific test.
The results are not ambiguous. They are not mixed. They are, in my view, one of the clearest clinical trial findings in recent cardiovascular medicine: not one of the six supplements significantly lowered LDL cholesterol compared to a sugar pill. Meanwhile, a single low dose of rosuvastatin lowered LDL by 38% in 28 days.
Every patient who is taking supplements instead of a statin needs to read this.
Heart Supplement Infographic Summary:

The Trial: SPORT, Supplements Placebo or Rosuvastatin Study
The study was conducted at the Cleveland Clinic between May 2021 and July 2022. Investigators enrolled 199 adults aged 40 to 75 years who met very specific criteria: they had an LDL-C between 70 and 189 mg/dL, no prior history of cardiovascular disease, and a 10-year ASCVD risk score between 5% and 20%, the range where guidelines currently recommend discussing statin therapy for primary prevention. They were not already on statins or lipid-lowering medications.
These were people who genuinely needed lipid management and were candidates for pharmacotherapy. They represent the exact patient population that is most likely to be tempted by supplement alternatives.
Participants were randomly assigned in equal numbers to one of eight groups:
- Rosuvastatin 5 mg daily (a low-dose, moderate-intensity statin)
- Placebo (matched in appearance to rosuvastatin)
- Fish oil (Nature Made brand, 2,400 mg daily)
- Cinnamon (NutriFlair brand, 2,400 mg daily)
- Garlic (Garlique brand, providing 5,000 mcg of allicin daily)
- Turmeric/Curcumin (BioSchwartz brand with bioperine, 4,500 mg daily)
- Plant Sterols (Nature Made CholestOff Plus, 1,600 mg daily)
- Red Yeast Rice (Arazo Nutrition brand, 2,400 mg daily)
The doses were chosen based on package labeling recommendations, using popular brands available in mainstream retail settings. This is critically important: these were not obscure or substandard products. These were exactly what a motivated, health-conscious consumer would buy at a pharmacy or order online.
Each participant had a fasting lipid panel and high-sensitivity C-reactive protein (hsCRP) measured at baseline and again at 28 days. The supplement bottles were returned for pill counts to verify adherence, which was high: fewer than 4 participants across all groups took less than 70% of their assigned regimen.
The primary endpoint was the percent change in LDL cholesterol after 28 days for rosuvastatin compared to placebo and each supplement in a prespecified hierarchical order.
Who Were the Participants?
The average participant was 64.4 years old. About 59% were women. The mean baseline LDL-C was 128.2 mg/dL. Median 10-year ASCVD risk was 7.9%. Hypertension was present in 42%, and about a third were former smokers. None had established cardiovascular disease.
This is a real, recognizable primary prevention population. These are the patients sitting in waiting rooms across America right now, some of whom are taking supplements because they are not sure they need or want a statin.
An important note on demographics: 89% of participants were non-Hispanic White. This is a meaningful limitation when generalizing the findings to more diverse populations, and the authors acknowledge it. The underlying pharmacology of these supplements and statins is not expected to differ by race, but the study cannot confirm that.
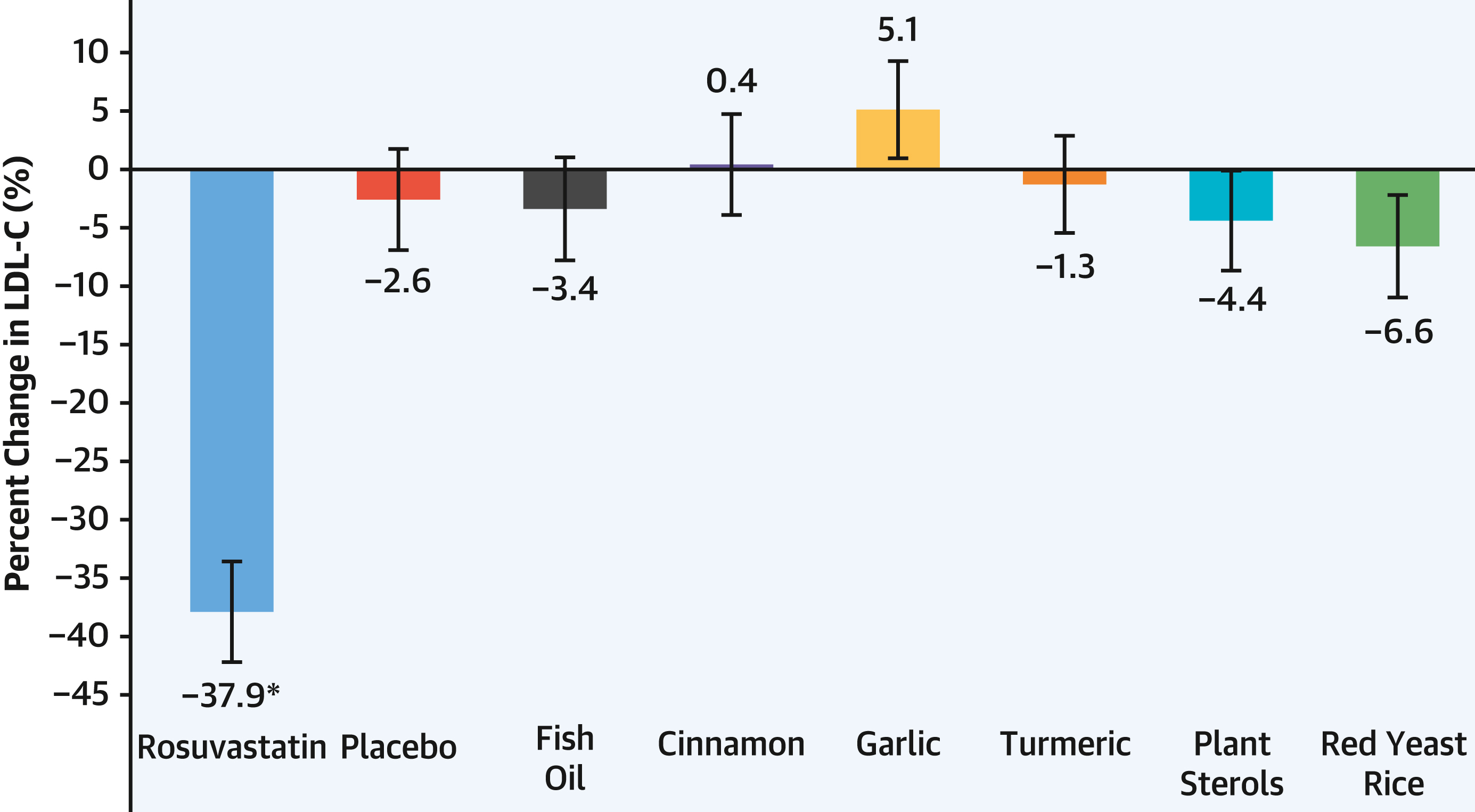
The Results: A Complete Scorecard
Here is how every arm performed:
|
Treatment |
LDL-C Change |
vs. Placebo |
vs. Rosuvastatin |
Verdict |
|
Rosuvastatin 5 mg |
-37.9% |
-35.2% (P<0.001) |
Reference |
WORKS |
|
Placebo |
-2.6% |
Reference |
+35.2% worse (P<0.001) |
Baseline |
|
Fish Oil |
-3.4% |
NS (P=0.80) |
+34.4% worse (P<0.001) |
FAILS |
|
Cinnamon |
+0.4% |
NS (P=0.33) |
+38.3% worse (P<0.001) |
FAILS |
|
Garlic |
+5.1% |
+7.8% INCREASE (P=0.01) |
+43.0% worse (P<0.001) |
HARMFUL |
|
Turmeric |
-1.3% |
NS (P=0.66) |
+36.6% worse (P<0.001) |
FAILS |
|
Plant Sterols |
-4.4% |
NS (P=0.58) |
+33.5% worse (P<0.001) |
FAILS |
|
Red Yeast Rice |
-6.6% |
NS (P=0.21) |
+31.3% worse (P<0.001) |
FAILS |
Let me walk through these results in detail because the numbers deserve full attention.
Rosuvastatin 5 mg: the medicine worked exactly as expected.
In just 28 days, at the lowest available dose of this medication, rosuvastatin reduced LDL-C by 37.9%. Every single participant randomized to rosuvastatin experienced a decrease in LDL-C. At least 18% and in half the participants, the reduction exceeded 40%. Total cholesterol dropped by 24.4% and triglycerides fell by 19.3%. This is a moderate-intensity statin doing what moderate-intensity statins do: reliably, predictably, and safely reducing atherogenic lipid burden.
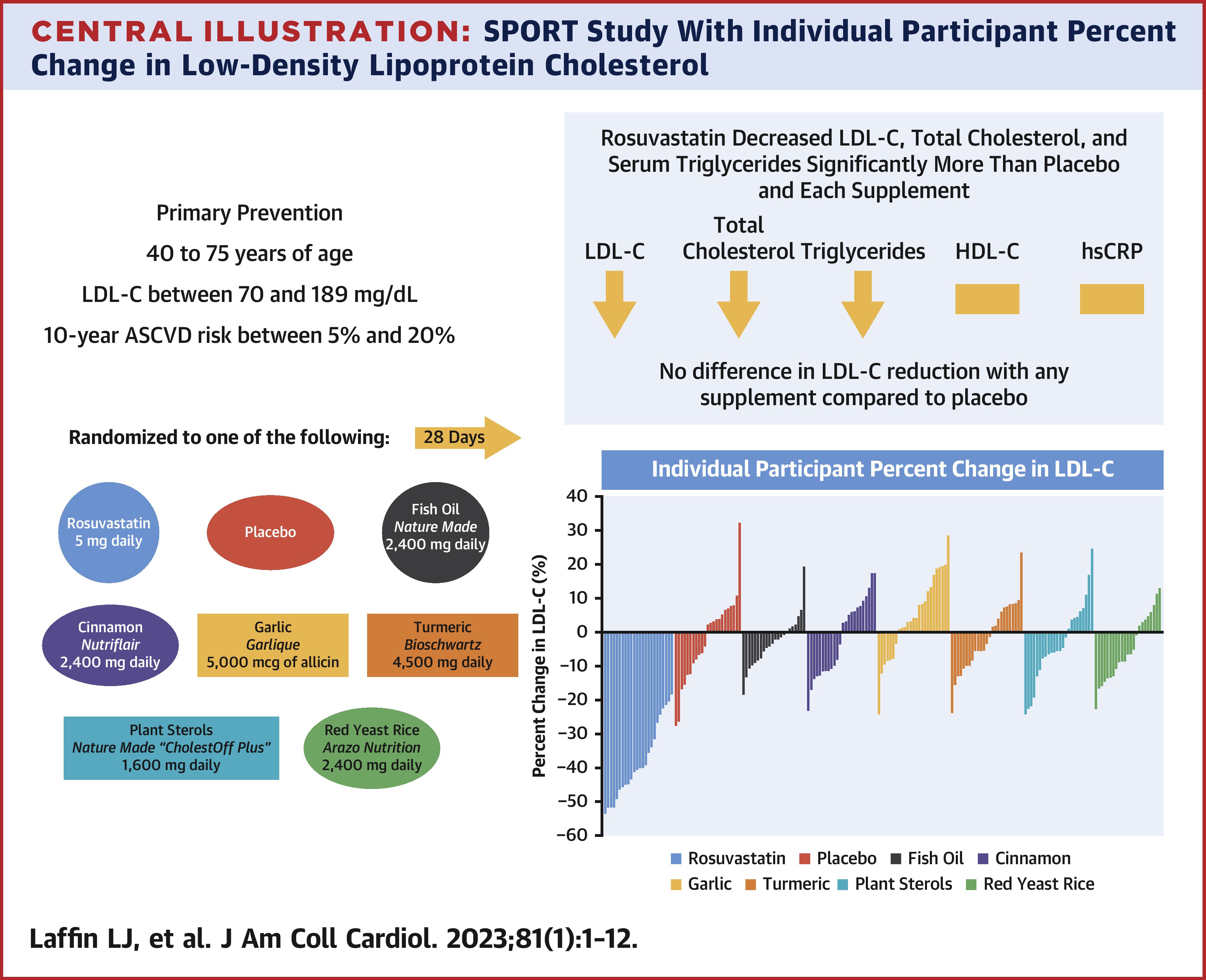
None of the supplements lowered LDL-C compared to placebo.
Read that again. Not one supplement in this trial, at commercially recommended doses, from well-known retail brands, produced a statistically significant reduction in LDL-C compared to a sugar pill over 28 days.
The numbers for the supplement arms were telling. Fish oil produced a 3.4% LDL reduction, not meaningfully different from the 2.6% placebo effect. Cinnamon produced a 0.4% increase. Turmeric produced a 1.3% decrease. Plant sterols produced a 4.4% decrease. Red yeast rice produced a 6.6% decrease. None of these reached statistical significance versus placebo.
Garlic actually raised LDL-C.
This is a finding that deserves emphasis. Participants assigned to garlic supplementation showed a 5.1% increase in LDL-C, which translated to a statistically significant increase of 7.8% compared to placebo (P=0.01). This is not a neutral result. This is a supplement that, in this rigorously controlled trial, moved the most important atherogenic biomarker in the wrong direction.
Prior data had suggested garlic might have a neutral effect on LDL-C. This trial found it may actually worsen the lipid profile. It is one thing for a supplement to not help. It is another for it to potentially harm.
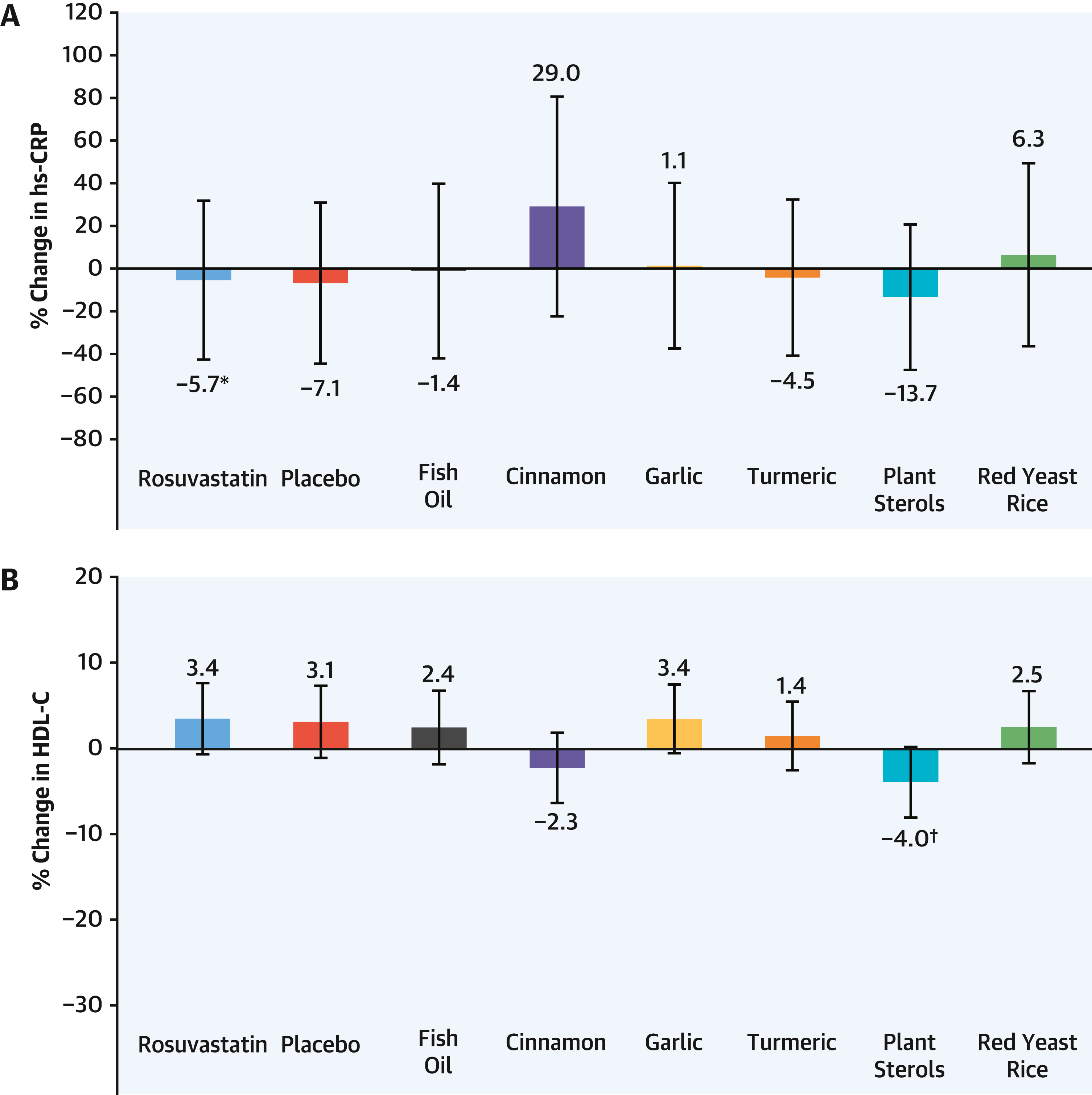
The secondary endpoints confirmed the pattern across the board.
|
Biomarker |
Rosuvastatin |
Best Supplement |
Difference |
P Value |
|
Total Cholesterol |
-24.4% |
Plant Sterols -3.9% |
~20 percentage points |
P<0.001 all comparisons |
|
Triglycerides |
-19.3% |
Fish Oil -5.0% |
~14 percentage points |
P<0.05 all comparisons |
|
HDL Cholesterol |
+3.4% |
No supplement improved HDL vs. rosuvastatin |
Plant sterols actually lowered HDL by 7.1% vs. placebo |
P=0.02 for plant sterols |
|
hsCRP (Inflammation) |
-5.7% |
Not significant for any arm |
No significant differences |
NS all |
One additional finding worth highlighting: plant sterols lowered HDL cholesterol compared to both rosuvastatin (by 7.5%, P=0.01) and compared to placebo (by 7.1%, P=0.02). A supplement that is marketed for heart health produced a significant reduction in the protective lipoprotein. This is the kind of finding that never appears on a supplement label.
Inflammation, as measured by hsCRP, was not significantly changed by any treatment arm, including rosuvastatin. This is slightly inconsistent with prior data showing statins reduce hsCRP, but the authors note this is likely a reflection of the small sample size and low dose used in SPORT. The JUPITER trial, which enrolled 17,802 patients on higher-dose rosuvastatin, showed robust CRP reductions.
Was It Safe? Yes, and That Is the Point About Statins
Adverse event rates were similar across all groups. Rosuvastatin produced no significant changes in liver enzymes, kidney function, or blood glucose over 28 days. There were no statin-related serious adverse events.
Two serious adverse events occurred in the trial: one deep vein thrombosis in the placebo group, and one liver adenocarcinoma discovered in the fish oil group. The liver cancer patient had not yet taken any study medication at the time of diagnosis, so it was clearly a pre-existing condition unmasked during screening. Neither event was related to the assigned treatment.
Numerically, adverse event rates were higher in the plant sterols and red yeast rice groups than in the rosuvastatin group. 28% of plant sterol participants and 28% of red yeast rice participants had adverse events, compared to 16% in the rosuvastatin and placebo groups. This did not reach statistical significance, but it is worth noting that these supplement arms were not demonstrably safer in clinical experience than the pharmaceutical option.
The statin safety concern is one of the most powerful drivers of supplement use among my patients. The fear of liver damage, muscle problems, and neurological side effects from statins is widespread, amplified by social media misinformation and wellness influencers who have built brands on statin skepticism. SPORT adds to a large and consistent body of evidence that low-dose rosuvastatin, and statins broadly, have an excellent short-term safety profile in primary prevention patients.
The Scam-Busting Reality: What This Trial Confirms About the Supplement Industry
The supplement industry in the United States operates in a regulatory environment that is fundamentally different from the one governing prescription medications. The Dietary Supplement Health and Education Act of 1994 (DSHEA) classifies supplements as foods, not drugs. The consequence of this classification is profound: supplement manufacturers are not required to prove their products work before selling them. The burden falls on the FDA to prove a product is unsafe, after it is already on the market.
The result? A market of over 90,000 products, generating nearly $50 billion in annual US sales, operating largely on marketing claims, consumer beliefs, and regulatory goodwill. By 2019, 77% of American adults reported taking dietary supplements. Of those, 18% said they were using supplements to promote heart health.
Consumer research cited in the SPORT paper reveals the core problem: most American consumers believe cholesterol health supplements are safer than prescription medications. And a majority believe they are equally or more effective than statins. Both beliefs are factually wrong, and the SPORT trial puts hard numbers on the wrongness.
This is not a minor misconception. When someone with an LDL of 140, a family history of premature coronary disease, hypertension, and a 12% 10-year risk score declines a statin because they are taking fish oil, they are making a medical decision based on false information. That decision, multiplied by millions of patients, translates into preventable heart attacks and strokes.
A note on red yeast rice that requires specific attention.
Red yeast rice contains monacolin K, which is chemically identical to lovastatin, a prescription statin. When red yeast rice does lower LDL-C in some studies, it is essentially acting as an unregulated, variable-dose statin. The FDA has intermittently taken enforcement action against red yeast rice products containing significant amounts of monacolin K, but enforcement is inconsistent.
The result is a supplement with variable potency, no standardized manufacturing quality, unknown monacolin K content, and no requirement to disclose its active pharmaceutical ingredient. In the SPORT trial, the specific Arazo Nutrition red yeast rice product produced a 6.6% LDL reduction, not significantly different from placebo. Different formulations might perform differently, but this variability is exactly the problem. You cannot know what you are getting, at what dose, or whether it is safe relative to known statin-drug interactions.
What about plant sterols? The European guidelines endorse them.
This is true. The 2019 ESC/EAS guidelines for dyslipidemia management mention plant sterols as an option for patients unwilling or unable to take statins. But the SPORT data are a reminder that endorsement in guidelines and efficacy in a rigorous head-to-head trial are different things. In SPORT, plant sterols showed a 4.4% LDL reduction that did not reach statistical significance compared to placebo, and notably reduced HDL cholesterol compared to both placebo and rosuvastatin.
There is also an ongoing scientific debate about whether plant sterols, which are absorbed into the bloodstream, might themselves be atherogenic. The SPORT trial was not designed or powered to evaluate cardiovascular outcomes, so it cannot answer that question. But it contributes another data point suggesting that plant sterols are not the neutral, beneficial alternative the marketing implies.
Why Patients Choose Supplements Over Statins: Understanding the Psychology
Understanding why so many patients reach for supplements instead of statins is essential for having productive conversations in the clinic. The SPORT paper identifies several drivers:
- Beliefs about statin safety.
Fear of hepatotoxicity is pervasive, despite the fact that clinically significant statin-induced liver injury is exceedingly rare and the FDA actually removed the requirement for routine liver enzyme monitoring from statin labels in 2012 because the data did not support it. Fear of muscle problems is more nuanced: statin-associated myalgia is real but often manageable, and serious rhabdomyolysis is rare, particularly at low and moderate doses.
- Beliefs about statin effectiveness relative to "natural" alternatives.
The word "natural" is one of the most powerful in consumer marketing. It implies safety, gentleness, and alignment with the body's own processes. Statins are synthetic molecules. Fish oil comes from fish. Cinnamon comes from a tree. The natural origin of a substance tells us nothing about its efficacy or safety, but it is a compelling heuristic for patients who are already skeptical of the pharmaceutical industry.
- Statin misinformation.
Dr. Nissen himself has written about what he calls the "statin denial cult," an internet-driven movement that attributes a vast range of symptoms and diseases to statins. These claims circulate on health blogs, social media, and podcasts and are extraordinarily difficult to counter because they tap into broader skepticism of pharmaceutical industry motives. The people spreading statin misinformation are often charismatic, credible-sounding, and deeply committed to their beliefs. The people publishing randomized clinical trials are not typically household names.
- The supplement industry's marketing infrastructure.
Supplement companies spend enormous sums on advertising that stops just short of making explicit drug claims. Phrases like "supports heart health," "promotes healthy cholesterol already in the normal range," and "cardiovascular support formula" are carefully calibrated to suggest benefit without triggering FDA drug claim enforcement. Consumers hear these phrases and make the inference the marketers intend: that the product helps their heart. The SPORT trial is now the clearest available evidence that for LDL-C, one of the most important modifiable cardiovascular risk factors, those implications are false.
Study Limitations to Be Honest About
The SPORT investigators are admirably transparent about the limitations of their trial, and intellectual honesty requires that I address them.
The trial duration was only 28 days.
This is long enough to see a statin's LDL-lowering effect, which typically plateaus within 2 to 4 weeks. But some supplements are hypothesized to work through mechanisms that require longer exposure: cumulative changes in gut microbiome composition, gradual modulation of enzyme pathways, or anti-inflammatory effects that unfold over months. The authors chose 28 days based on guideline recommendations for reassessing lipid response to therapy. It is possible, though not established, that some supplements might show modest effects over longer periods. However, there is no strong prior evidence suggesting that the supplements tested here show substantially better results at 3 or 6 months compared to 4 weeks.
The sample size was small.
Twenty-five participants per group is sufficient to detect a 15% difference in LDL-C between rosuvastatin and any supplement with 90% statistical power, and the trial delivered that. But it is not large enough to rule out small benefits from any individual supplement, to conduct meaningful subgroup analyses, or to evaluate cardiovascular outcomes. For outcome data, we would need tens of thousands of patients followed for years. SPORT was not that trial, and it did not claim to be.
The population was predominantly non-Hispanic White.
89% non-Hispanic White is not representative of the diverse American population that uses supplements for cholesterol management. Whether these results generalize to Black, Hispanic, or Asian American patients cannot be confirmed from this dataset.
There was no formal blinding of participants.
Because the supplements came in different numbers and shapes of pills, full blinding was not possible. Investigators were blinded, the event adjudication committee was blinded, and the supplements were repackaged in unlabeled bottles. But participants could potentially infer their assignment. This is a practical limitation of supplement trial design, not a flaw in execution, and it does not explain the complete absence of LDL-C benefit in any supplement arm.
The trial was funded by AstraZeneca.
AstraZeneca manufactures rosuvastatin (Crestor). The study was investigator-initiated and funded by an unrestricted grant. The sponsor had no role in trial design, data collection, analysis, interpretation, or the decision to publish, according to the authors' attestation. The statistical analysis was performed by the C5Research coordinating center, not the sponsor. These protections are meaningful, but intellectual honesty requires disclosing the funding relationship and letting readers weigh it.
My Clinical Perspective: What I Tell My Patients
I have this conversation regularly. A patient with an LDL of 135, blood pressure of 138/88, a father who had a heart attack at 58, and a 10-year risk score of 11% tells me they are taking fish oil and red yeast rice and doing not need a statin. Here is how I approach that conversation, informed by what the evidence, including SPORT, actually shows:
I start with the numbers.
LDL causes atherosclerosis. This is not a hypothesis. It is established causally by genetic studies, Mendelian randomization, and decades of randomized trial data. The Ference et al. European Atherosclerosis Society consensus statement established this beyond reasonable scientific doubt. Every 39 mg/dL reduction in LDL-C reduces cardiovascular events by roughly 22% over 5 years. At a modest statin dose of 5 mg rosuvastatin, we can drop LDL by nearly 40% in a month. Your supplements dropped it by less than a placebo in the only rigorous trial that has ever compared them.
I address the safety concerns directly.
Statin-related serious muscle injury (rhabdomyolysis) is rare, occurring in fewer than 1 in 100,000 patients on standard doses. Clinically significant liver injury is even rarer. New-onset diabetes is a real, small but real, risk from statins, particularly at higher doses in patients already at metabolic risk. These are real tradeoffs worth discussing. But none of the supplement arms in SPORT were demonstrably safer than rosuvastatin in clinical experience, and two supplement arms had numerically higher adverse event rates. The safety advantage of supplements is assumed, not proven.
I clarify what fish oil actually does.
Fish oil at high doses, specifically prescription-strength omega-3 fatty acids such as icosapentaenoic acid (EPA) alone, does have cardiovascular outcome data from the controversial REDUCE-IT trial showing a 25% reduction in cardiovascular events. But that was 4 grams daily of a specific, highly purified EPA formulation in patients already on statins with elevated triglycerides and established cardiovascular disease or diabetes. That is a profoundly different patient and product from the 2,400 mg of over-the-counter Nature Made fish oil tested in SPORT, which was not used in combination with a statin and produced no meaningful LDL-C effect. Conflating these is a common source of confusion that supplements marketing has been happy to exploit. The REDUCE-IT trial is controversial because the placebo was an inflammatory placebo. I personally do not recommend EPA nor Vascepa. Fish oil also has concerns for higher incidence of atrial fibrillation.
I address the 'natural' argument.
Arsenic is natural. Cyanide is natural. Botulinum toxin, the most acutely lethal substance known to science, is natural. Naturalness tells us nothing about safety or efficacy. Conversely, rosuvastatin, which is synthetic, has been studied in more than 170,000 randomized trial participants with a safety profile that has been validated over decades. The question is not 'is it natural?' The question is 'does it work, and is it safe?' On both counts, statins win and supplements lose.
I do not dismiss patient autonomy.
Ultimately, a patient who understands the evidence and still prefers not to take a statin has the right to make that choice. My job is to make sure they are making it with accurate information. After the SPORT conversation, I want my patient to understand that their fish oil is probably not doing anything meaningful for their LDL, that their garlic supplement might actually be raising it, and that the statin they have been avoiding is safe, affordable, and effective at a level no supplement has ever demonstrated in a rigorous trial.
The Bottom Line
The SPORT trial is the most important supplement study published in cardiovascular medicine in years. In a well-designed, randomized, placebo-controlled trial of the six most popular cholesterol supplements at full commercial doses, over 28 days, in a real-world primary prevention population:
- Not one supplement significantly lowered LDL-C compared to placebo.
- Garlic raised LDL-C significantly compared to placebo.
- Plant sterols lowered HDL-C, the protective cholesterol, compared to both placebo and rosuvastatin.
- Rosuvastatin 5 mg lowered LDL-C by 38%, total cholesterol by 24%, and triglycerides by 19%, significantly better than every comparator, without meaningful adverse effects.
- The difference in LDL-C reduction between rosuvastatin and placebo was 35.2 percentage points. The difference between rosuvastatin and the best-performing supplement (red yeast rice at -6.6%) was still over 31 percentage points.
This is not a close call. This is not a finding that requires nuanced interpretation or caveat-heavy hedging. The supplements marketed to Americans for cholesterol health do not lower cholesterol compared to placebo. The medication they are being used to replace lowers it by nearly 40% in a month.
If you are taking cholesterol supplements in place of a medication your doctor has recommended, please have an honest conversation with your cardiologist about what the evidence actually shows. Bring this paper. Bring your lipid numbers. Bring your honest questions about statin safety.
Because the only thing more expensive than a statin copay is a heart attack.
Reference
Laffin LJ, Bruemmer D, Garcia M, et al. Comparative Effects of Low-Dose Rosuvastatin, Placebo, and Dietary Supplements on Lipids and Inflammatory Biomarkers. J Am Coll Cardiol. 2023;81(1):1-12. doi:10.1016/j.jacc.2022.10.013 https://www.jacc.org/doi/10.1016/j.jacc.2022.10.013
Still Have Questions? Stop Googling and Ask Dr. Alo.
You’ve read the science, but applying it to your own life can be confusing. I created the Dr. Alo VIP Private Community to be a sanctuary away from social media noise.
Inside, you get:
-
Direct Access: I answer member questions personally 24/7/365.
-
Weekly Live Streams: Deep dives into your specific health challenges.
-
Vetted Science: No fads, just evidence-based cardiology and weight loss.
Don't leave your heart health to chance. Get the guidance you deserve. All this for less than 0.01% the cost of health insurance! You can cancel at anytime!
[👉 Join the Dr. Alo VIP Community Today]




















